
Summary
Berotralstat, sold under the brand name Orladeyo, is a medication used to prevent attacks of hereditary angioedema (HAE) in people aged twelve years and older.[3][5][7][8][4]
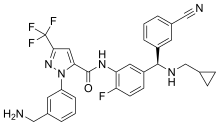 | |
| Clinical data | |
|---|---|
| Trade names | Orladeyo |
| Other names | BCX7353, BCX-7353 |
| License data | |
| Routes of administration | By mouth |
| Drug class | Plasma kallikrein inhibitor |
| ATC code |
|
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID |
|
| DrugBank |
|
| ChemSpider |
|
| UNII |
|
| KEGG |
|
| Chemical and physical data | |
| Formula | C30H26F4N6O |
| Molar mass | 562.573 g·mol−1 |
| 3D model (JSmol) |
|
| |
| |
The most common side effects include abdominal pain, vomiting, diarrhea, back pain, and heartburn.[4]
Berotralstat was approved for medical use in the United States in December 2020,[3][4][9] and in the European Union in April 2021.[5]
History edit
Berotralstat was approved based on evidence from one clinical trial (Trial 1 /NCT03485911) of 120 participants with hereditary angioedema.[4] The trial was conducted at 40 sites in the United States, the European Union, and Canada.[4] Trial investigators evaluated participants 12 years and older[10] with hereditary angioedema for eight weeks to determine the number of attacks for each participant.[4] The trial enrolled only participants who had at least two attacks during the eight-week period.[4] Participants were assigned to receive one of two doses of berotralstat or placebo once every day for 24 weeks.[4] Neither the participants nor the investigators knew which treatment was being given until after the trial was completed.[4] All participants could use other medications for treatment of attacks.[4]
References edit
- ^ "Summary Basis of Decision - Orladeyo". Health Canada. 23 October 2014. Retrieved 28 November 2022.
- ^ Product monograph hres.ca
- ^ a b c "Orladeyo- berotralstat hydrochloride capsule". DailyMed. Retrieved 25 December 2020.
- ^ a b c d e f g h i j k "Drug Trials Snapshot: Orladeyo". U.S. Food and Drug Administration. 3 December 2020. Retrieved 25 December 2020. This article incorporates text from this source, which is in the public domain.
- ^ a b c "Orladeyo EPAR". European Medicines Agency (EMA). 24 February 2021. Retrieved 12 July 2021.
- ^ "Orladeyo Product information". Union Register of medicinal products. Retrieved 3 March 2023.
- ^ Hwang JR, Hwang G, Johri A, Craig T (December 2019). "Oral plasma kallikrein inhibitor BCX7353 for treatment of hereditary angioedema". Immunotherapy. 11 (17): 1439–1444. doi:10.2217/imt-2019-0128. PMID 31635497.
- ^ Zuraw B, Lumry WR, Johnston DT, Aygören-Pürsün E, Banerji A, Bernstein JA, et al. (October 2020). "Oral once-daily berotralstat for the prevention of hereditary angioedema attacks: A randomized, double-blind, placebo-controlled phase 3 trial". The Journal of Allergy and Clinical Immunology. 148 (1): 164–172.e9. doi:10.1016/j.jaci.2020.10.015. PMID 33098856.
- ^ "Orladeyo: FDA-Approved Drugs". U.S. Food and Drug Administration (FDA). Retrieved 25 December 2020.
- ^ "Berotralstat (Oral Route) Side Effects - Mayo Clinic". www.mayoclinic.org. Retrieved 3 March 2021.
External links edit
- "Berotralstat". Drug Information Portal. U.S. National Library of Medicine.
- Clinical trial number NCT03485911 for "Efficacy and Safety Study of BCX7353 as an Oral Treatment for the Prevention of Attacks in HAE (APeX-2)" at ClinicalTrials.gov


