
Summary
Calcipotriol, also known as calcipotriene, is a synthetic derivative of calcitriol, a form of vitamin D.[medical citation needed] It is used in the treatment of psoriasis.[medical citation needed] It is safe for long-term application in psoriatic skin conditions.[medical citation needed]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Daivonex, Dovonex, Sorilux |
| Other names | calcipotriene (USAN US) |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a608018 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | Topical administration |
| ATC code |
|
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 5 to 6% |
| Metabolism | Liver |
| Excretion | Biliary |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID |
|
| IUPHAR/BPS |
|
| DrugBank |
|
| ChemSpider |
|
| UNII |
|
| KEGG |
|
| ChEBI |
|
| ChEMBL |
|
| CompTox Dashboard (EPA) |
|
| ECHA InfoCard | 100.119.473 |
| Chemical and physical data | |
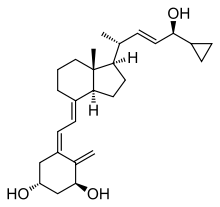
| Formula | C27H40O3 |
| Molar mass | 412.614 g·mol−1 |
| 3D model (JSmol) |
|
| |
| |
| | |
It was patented in 1985 and approved for medical use in 1991.[1] It is marketed under the trade name "Dovonex" in the United States, "Daivonex" outside North America, and "Psorcutan" in Germany.[citation needed]
It is on the World Health Organization's List of Essential Medicines.[2]
Calcipotriol is also available as Calcipotriol/betamethasone dipropionate, a fixed-dose combination medication with the synthetic corticosteroid betamethasone dipropionate for the treatment of plaque psoriasis.[3]
Medical uses edit
Chronic plaque psoriasis is the chief medical use of calcipotriol.[4] It has also been used successfully in the treatment of alopecia areata.[5]
Contraindications edit
Hypersensitivity, use on face, hypercalcaemia, or evidence of vitamin D toxicity are the only contraindications for calcipotriol use.[6]
Cautions include exposure to excessive natural or artificial light, due to the potential for calcipotriol to cause photosensitivity.[6]
Adverse effects edit
Adverse effects by frequency:[4][6][7][8]
- Very common (> 10% frequency)
- Burning
- Itchiness
- Skin irritation
- Common (1–10% frequency)
- Dermatitis
- Dry skin
- Erythema
- Peeling
- Worsening of psoriasis including facial/scalp
- Rash
- Uncommon (0.1–1% frequency)
- Exacerbation of psoriasis
- Rare (< 0.1% frequency)
- Allergic contact dermatitis
- Hypercalcaemia
- Photosensitivity
- Changes in pigmentation
- Skin atrophy
Interactions edit
No drug interactions are known.[6]
Pharmacology edit
Mechanism of action edit
The efficacy of calcipotriol in the treatment of psoriasis was first noticed by the observation of patients receiving various forms of vitamin D in an osteoporosis study. Unexpectedly, some patients who also had psoriasis experienced dramatic reductions in lesion counts.[9]
The precise mechanism of calcipotriol in remitting psoriasis is not well understood. However, it has been shown to have comparable affinity with calcitriol for the vitamin D receptor (VDR), while being less than 1% as active as the calcitriol in regulating calcium metabolism. The vitamin D receptor belongs to the steroid/thyroid receptor superfamily, and is found on the cells of many different tissues including the thyroid, bone, kidney, and T cells of the immune system. T cells are known to play a role in psoriasis, and it is thought that the binding of calcipotriol to the VDR modulates the T cells gene transcription of cell differentiation and proliferation related genes.
In mouse studies, topical calcipotriol administration to the ear and dorsal skin led to a dose-dependent increase in the production of the epithelial cell-derived cytokine TSLP by keratinocytes, and triggered atopic dermatitis at high concentrations.[10] This upregulation of TSLP production due to calcipotriol application is thought to be mediated through the coactivation of vitamin D receptor/RXRα and vitamin D receptor/RXRβ heterodimers. As psoriasis is typically thought to be partially driven by Th1/Th17 inflammatory cytokines,[11] calcipotriol treatment at appropriate concentrations may alleviate psoriasis symptoms by repressing Th1/Th17 inflammation through TSLP production, which is linked to a Th2 response. However, it is important to note that this has not yet been confirmed.
Pharmacokinetics edit
After application and systemic uptake, calcipotriol undergoes rapid hepatic metabolism. Calcipotriol is metabolized to MC1046 (the α,β−unsaturated ketone analog), which is subsequently metabolized to its primary metabolite, the saturated ketone analog MC1080. MC1080 is then slowly metabolized to calcitroic acid.[12]
The metabolites of calcipotriol are less potent than the parent compound.
Chemistry edit
Calcipotriol is a white to almost white crystalline compound.
References edit
- ^ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 452. ISBN 9783527607495.
- ^ World Health Organization (2021). World Health Organization model list of essential medicines: 22nd list (2021). Geneva: World Health Organization. hdl:10665/345533. WHO/MHP/HPS/EML/2021.02.
- ^ "Taclonex- calcipotriene and betamethasone dipropionate ointment". DailyMed. 21 May 2020. Retrieved 19 October 2020.
- ^ a b Rossi S, ed. (2013). Australian Medicines Handbook (2013 ed.). Adelaide: The Australian Medicines Handbook Unit Trust. ISBN 978-0-9805790-9-3.
- ^ Kim DH, Lee JW, Kim IS, Choi SY, Lim YY, Kim HM, et al. (August 2012). "Successful treatment of alopecia areata with topical calcipotriol". Annals of Dermatology. 24 (3): 341–344. doi:10.5021/ad.2012.24.3.341. PMC 3412244. PMID 22879719.
- ^ a b c d "Dovonex, Calcitrene Ointment (calcipotriene) dosing, indications, interactions, adverse effects, and more". Medscape Reference. WebMD. Retrieved 26 January 2014.
- ^ "CALCIPOTRIENE (calcipotriene) solution [E. FOUGERA & CO. A division of Fougera Pharmaceuticals Inc.]". DailyMed. E. FOUGERA & CO. A division of Fougera Pharmaceuticals Inc. May 2012. Retrieved 26 January 2014.
- ^ "PRODUCT INFORMATION DAIVONEX® CREAM Calcipotriol 50 microgram/g" (PDF). TGA eBusiness Services. LEO Pharma Pty Ltd. 28 April 2011. Retrieved 26 January 2014.
- ^ Morimoto S, Kumahara Y (March 1985). "A patient with psoriasis cured by 1 alpha-hydroxyvitamin D3". Medical Journal of Osaka University. 35 (3–4): 51–54. PMID 4069059.
- ^ Li M, Hener P, Zhang Z, Kato S, Metzger D, Chambon P (August 2006). "Topical vitamin D3 and low-calcemic analogs induce thymic stromal lymphopoietin in mouse keratinocytes and trigger an atopic dermatitis". Proceedings of the National Academy of Sciences of the United States of America. 103 (31): 11736–11741. Bibcode:2006PNAS..10311736L. doi:10.1073/pnas.0604575103. PMC 1544239. PMID 16880407.
- ^ Wong T, Hsu L, Liao W (2013-02-01). "Phototherapy in psoriasis: a review of mechanisms of action". Journal of Cutaneous Medicine and Surgery. 17 (1): 6–12. doi:10.2310/7750.2012.11124. PMC 3736829. PMID 23364144.
- ^ "Enstilar (calcipotriene and betamethasone dipropionate) Foam, 0.005%/0.064% for topical use. Full Prescribing Information" (PDF). Parsippany, NJ: LEO Pharma Inc. 2015. Archived from the original (PDF) on 2018-09-20. Retrieved 2015-11-21.
External links edit
- "Calcipotriene". Drug Information Portal. U.S. National Library of Medicine.


