
Summary
In organic chemistry, a carbamate is a category of organic compounds with the general formula R2NC(O)OR and structure >N−C(=O)−O−, which are formally derived from carbamic acid (NH2COOH). The term includes organic compounds (e.g., the ester ethyl carbamate), formally obtained by replacing one or more of the hydrogen atoms by other organic functional groups; as well as salts with the carbamate anion H2NCOO− (e.g. ammonium carbamate).[1]
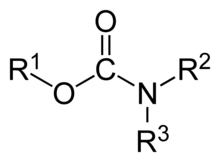
Polymers whose repeat units are joined by carbamate like groups −NH−C(=O)−O− are an important family of plastics, the polyurethanes. See Etymology for clarification.
Properties edit
While carbamic acids are unstable, many carbamate esters and salts are stable and well known.[2]
Equilibrium with carbonate and bicarbonate edit
In water solutions, the carbamate anion slowly equilibrates with the ammonium NH+
4 cation and the carbonate CO2−
3 or bicarbonate HCO−
3 anions:[3][4][5]
- H2NCO−2 + 2 H2O ⇌ NH+4 + HCO−3 + OH−
- H2NCO−2 + H2O ⇌ NH+4 + CO2−3
Calcium carbamate is soluble in water, whereas calcium carbonate is not. Adding a calcium salt to an ammonium carbamate/carbonate solution will precipitate some calcium carbonate immediately, and then slowly precipitate more as the carbamate hydrolyzes.[3]
Synthesis edit
Carbamate salts edit
The salt ammonium carbamate is generated by treatment of ammonia with carbon dioxide:[6]
- 2 NH3 + CO2 → NH4[H2NCO2]
Carbamate esters edit
Carbamate esters also arise via alcoholysis of carbamoyl chlorides:[1]
- R2NC(O)Cl + R'OH → R2NCO2R' + HCl
Alternatively, carbamates can be formed from chloroformates and amines:[7]
- R'OC(O)Cl + R2NH → R2NCO2R' + HCl
Carbamates may be formed from the Curtius rearrangement, where isocyanates formed are reacted with an alcohol.[7]
- RCON3 → RNCO + N2
- RNCO + R′OH → RNHCO2R′
Natural occurrence edit
Within nature carbon dioxide can bind with neutral amine groups to form a carbamate, this post-translational modification is known as carbamylation. This modification is known to occur on several important proteins; see examples below.[8]
Hemoglobin edit
The N-terminal amino groups of valine residues in the α- and β-chains of deoxyhemoglobin exist as carbamates. They help to stabilise the protein when it becomes deoxyhemoglobin, and increases the likelihood of the release of remaining oxygen molecules bound to the protein. This stabilizing effect should not be confused with the Bohr effect (an indirect effect caused by carbon dioxide).[9]
Urease and phosphotriesterase edit
The ε-amino groups of the lysine residues in urease and phosphotriesterase also feature carbamate. The carbamate derived from aminoimidazole is an intermediate in the biosynthesis of inosine. Carbamoyl phosphate is generated from carboxyphosphate rather than CO2.[10]
CO2 capture by ribulose 1,5-bisphosphate carboxylase edit
Perhaps the most prevalent carbamate is the one involved in the capture of CO2 by plants. This process is necessary for their growth. The enzyme ribulose 1,5-bisphosphate carboxylase/oxygenase (RuBisCO) fixes a molecule of carbon dioxide as phosphoglycerate in the Calvin cycle. At the active site of the enzyme, a Mg2+ ion is bound to glutamate and aspartate residues as well as a lysine carbamate. The carbamate is formed when an uncharged lysine side chain near the ion reacts with a carbon dioxide molecule from the air (not the substrate carbon dioxide molecule), which then renders it charged, and, therefore, able to bind the Mg2+ ion.[11]
Applications edit
Synthesis of urea edit
Although not usually isolated as such, the salt ammonium carbamate is produced on a large scale as an intermediate in the production of the commodity chemical urea from ammonia and carbon dioxide.[1]
Polyurethane plastics edit
Polyurethanes contain multiple carbamate groups as part of their structure. The "urethane" in the name "polyurethane" refers to these carbamate groups; the term "urethane links" describe how carbamates polymerize.[Requires Citation] In contrast, the substance commonly called "urethane", ethyl carbamate, is neither a component of polyurethanes, nor is it used in their manufacture.[Requires Citation] Urethanes are usually formed by reaction of an alcohol with an isocyanate.[Requires Citation] Commonly, urethanes made by a non-isocyanate route are called carbamates.[Requires Citation]
Polyurethane polymers have a wide range of properties and are commercially available as foams, elastomers, and solids. Typically, polyurethane polymers are made by combining diisocyanates, e.g. toluene diisocyanate, and diols, where the carbamate groups are formed by reaction of the alcohols with the isocyanates:[12]
- RN=C=O + R′OH → RNHC(O)OR′
Carbamate insecticides edit
The so-called carbamate insecticides feature the carbamate ester functional group. Included in this group are aldicarb (Temik), carbofuran (Furadan), carbaryl (Sevin), ethienocarb, fenobucarb, oxamyl, and methomyl. These insecticides kill insects by reversibly inactivating the enzyme acetylcholinesterase (AChE inhibition)[13] (IRAC mode of action 1a).[14] The organophosphate pesticides also inhibit this enzyme, although irreversibly, and cause a more severe form of cholinergic poisoning[15] (the similar IRAC MoA 1b).[14]
Fenoxycarb has a carbamate group but acts as a juvenile hormone mimic, rather than inactivating acetylcholinesterase.[16]
The insect repellent icaridin is a substituted carbamate.[17]
Besides their common use as arthropodocides/insecticides, they are also nematicidal.[18] One such is Oxamyl.[18]
Sales have declined dramatically over recent decades.[18]
Resistance edit
Among mutations in esterases, carbamate resistance most commonly involves acetylcholinesterase (AChE) desensitization, while organophosphate resistance most commonly is carboxylesterase metabolization.[19]
Carbamate nerve agents edit
While the carbamate acetylcholinesterase inhibitors are commonly referred to as "carbamate insecticides" due to their generally high selectivity for insect acetylcholinesterase enzymes over the mammalian versions, the most potent compounds such as aldicarb and carbofuran are still capable of inhibiting mammalian acetylcholinesterase enzymes at low enough concentrations that they pose a significant risk of poisoning to humans, especially when used in large amounts for agricultural applications. Other carbamate based acetylcholinesterase inhibitors are known with even higher toxicity to humans, and some such as T-1123 and EA-3990 were investigated for potential military use as nerve agents. However, since all compounds of this type have a quaternary ammonium group with a permanent positive charge, they have poor blood–brain barrier penetration, and also are only stable as crystalline salts or aqueous solutions, and so were not considered to have suitable properties for weaponisation.[20][21]
Preservatives and cosmetics edit
Iodopropynyl butylcarbamate is a wood and paint preservative and used in cosmetics.[22]
Chemical research edit
Some of the most common amine protecting groups, such as BOC,[23] FMOC,[24] benzyl chloroformate[25] and trichloroethyl chloroformate[26] are carbamates.
Medicine edit
Ethyl carbamate edit
Urethane (ethyl carbamate) was once produced commercially in the United States as a chemotherapy agent and for other medicinal purposes. It was found to be toxic and largely ineffective.[27] It is occasionally used in veterinary medicine in combination with other drugs to produce anesthesia.[28]
Carbamate drugs edit
In addition, some carbamates are used in human pharmacotherapy, for example, the acetylcholinesterase inhibitors neostigmine and rivastigmine, whose chemical structure is based on the natural alkaloid physostigmine. Other examples are meprobamate and its derivatives like carisoprodol, felbamate, mebutamate, phenprobamate, and tybamate, a class of anxiolytic and muscle relaxant drugs widely used in the 1960s before the rise of benzodiazepines, and still used nowadays in some cases. Carbachol is primarily used for various ophthalmic purposes.[29]
The protease inhibitor darunavir for HIV treatment also contains a carbamate functional group.[30]
Ephedroxane, an aminorex analogue used as a stimulant, also falls into the carbamate category.[31]
Toxicity edit
Besides inhibiting human acetylcholinesterase[32] (although to a lesser degree than the insect enzyme), carbamate insecticides also target human melatonin receptors.[33] The human health effects of carbamates are well documented in the list of known endocrine disruptor compounds.[34] Clinical effects of carbamate exposure can vary from slightly toxic to highly toxic depending on a variety of factors including such as dose and route of exposure with ingestion and inhalation resulting in the most rapid clinical effects.[34] These clinical manifestations of carbamate intoxication are muscarinic signs, nicotinic signs, and in rare cases central nervous system signs.[34]
Sulfur analogues edit
There are two oxygen atoms in a carbamate (1), ROC(=O)NR2, and either or both of them can be conceptually replaced by sulfur. Analogues of carbamates with only one of the oxygens replaced by sulfur are called thiocarbamates (2 and 3). Carbamates with both oxygens replaced by sulfur are called dithiocarbamates (4), RSC(=S)NR2.[35]
There are two different structurally isomeric types of thiocarbamate:
- O-thiocarbamates (2), ROC(=S)NR2, where the carbonyl group (C=O) is replaced with a thiocarbonyl group (C=S)[36]
- S-thiocarbamates (3), RSC(=O)NR2, where the R–O– group is replaced with an R–S– group[36]
O-thiocarbamates can isomerise to S-thiocarbamates, for example in the Newman–Kwart rearrangement.[37]
Etymology edit
The etymology of the words "urethane" and "carbamate" are highly similar but not the same. The word "urethane" was first coined in 1833 by French chemist Jean-Baptiste Dumas.[38][39] Dumas states "Urethane. The new ether, brought into contact with liquid and concentrated ammonia, exerts on this substance a reaction so strong that the mixture boils, and sometimes even produces a sort of explosion. If the ammonia is in excess, all the ether disappears. It forms ammonium hydrochlorate and a new substance endowed with interesting properties."[39] Dumas appears to be naming this compound urethane. However, later Dumas states "While waiting for opinion to settle on the nature of this body, I propose to designate by the names of urethane and oxamethane the two materials which I have just studied, and which I regard as types of a new family, among nitrogenous substances. These names which, in my eyes, do not prejudge anything in the question of alcohol and ethers, will at least have the advantage of satisfying chemists who still refuse to accept our theory."[39] The word urethane is derived from the words "urea" and "ether" with the suffix "-ane" as a generic chemical suffix, making it specific for the R2NC(=O)OR' (R' not = H) bonding structure.[40]
The use of the word "carbamate" appears to come later only being traced back to at least 1849, in a description of Dumas's work by Henry Medlock.[41] Medlock states "It is well known that the action of ammonia on chloro-carbonate (phosgene) of ethyl gives rise to the formation of the substance which Dumas, the discoverer, called urethane, and which we are now in the habit of considering as the ether of carbamic acid."[41] This suggests that instead of continuing with the urethane family naming convention Dumas coined, they altered the naming convention to ethyl ether of carbamic acid. Carbamate is derived from the words "carbamide", otherwise known as urea, and "-ate" a suffix which indicates the salt or ester of an acid.[42][43]
Both words have roots deriving from urea. Carbamate is less-specific because the -ate suffix is ambiguous for either the salt or ester of a carbamic acid. However, the -ate suffix is also more specific because it suggests carbamates must be derived from the acid of carbamate, or carbamic acids. Although, a urethane has the same chemical structure as a carbamate ester moiety, a urethane not derived from a carbamic acid is not a carbamate ester. In other words, any synthesis of the R2NC(=O)OR' (R' not = H) moiety that does not derive from carbamic acids is not a carbamate ester but instead a urethane. Furthermore, carbamate esters are urethanes but not all urethanes are carbamate esters. This further suggests that polyurethanes are not simply polycarbamate-esters because polyurethanes are not typically synthesized using carbamic acids.
IUPAC states "The esters are often called urethanes or urethans, a usage that is strictly correct only for the ethyl esters."[44] But also states, "An alternative term for the compounds R2NC(=O)OR' (R' not = H), esters of carbamic acids, R,NC(=O)OH, in strict use limited to the ethyl esters, but widely used in the general sense".[45] IUPAC provides these statements without citation.
See also edit
References edit
- ^ a b c Jäger, Peter; Rentzea, Costin N.; Kieczka, Heinz (2000). "Carbamates and Carbamoyl Chlorides". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a05_051. ISBN 978-3527306732.
- ^ Ghosh, Arun K.; Brindisi, Margherita (2015-04-09). "Organic Carbamates in Drug Design and Medicinal Chemistry". Journal of Medicinal Chemistry. 58 (7): 2895–2940. doi:10.1021/jm501371s. ISSN 0022-2623. PMC 4393377. PMID 25565044.
- ^ a b Burrows, George H.; Lewis, Gilbert N. (1912). "The equilibrium between ammonium carbonate and ammonium carbamate in aqueous solution at 25°". Journal of the American Chemical Society. 34 (8): 993–995. doi:10.1021/ja02209a003.
- ^ Clark, K. G.; Gaddy, V. L.; Rist, C. E. (1933). "Equilibria in the Ammonium Carbamate-Urea-Water System". Ind. Eng. Chem. 25 (10): 1092–1096. doi:10.1021/ie50286a008.
- ^ Mani, Fabrizio; Peruzzini, Maurizio; Stoppioni, Piero (2006). "CO2 absorption by aqueous NH
3 solutions: speciation of ammonium carbamate, bicarbonate and carbonate by a 13
C NMR study". Green Chemistry. 8 (11). Royal Society of Chemistry: 995. doi:10.1039/b602051h. ISSN 1463-9262. - ^ Brooks, L. A.; Audrieta, L. F.; Bluestone, H.; Jofinsox, W. C. (1946). "Ammonium Carbamate". Inorganic Syntheses. Vol. 2. pp. 85–86. doi:10.1002/9780470132333.ch23. ISBN 978-0-470-13233-3.
- ^ a b Chaturvedi, Devdutt (1 May 2011). "Recent Developments on the Carbamation of Amines". Current Organic Chemistry. 15 (10). Bentham Science Publishers Ltd.: 1593–1624. doi:10.2174/138527211795378173. ISSN 1385-2728.
- ^ Linthwaite, Victoria L.; Janus, Joanna M.; Brown, Adrian P.; Wong-Pascua, David; O'Donoghue, AnnMarie C.; Porter, Andrew; Treumann, Achim; Hodgson, David R. W.; Cann, Martin J. (2018-08-06). "The identification of carbon dioxide mediated protein post-translational modifications". Nature Communications. 9 (1): 3092. Bibcode:2018NatCo...9.3092L. doi:10.1038/s41467-018-05475-z. ISSN 2041-1723. PMC 6078960. PMID 30082797.
- ^ Ferguson, J. K. W.; Roughton, F. J. W. (1934-12-14). "The direct chemical estimation of carbamino compounds of CO2 with hæmoglobin". The Journal of Physiology. 83 (1): 68–86. doi:10.1113/jphysiol.1934.sp003212. ISSN 0022-3751. PMC 1394306. PMID 16994615.
- ^ Bartoschek, S.; Vorholt, J. A.; Thauer, R. K.; Geierstanger, B. H.; Griesinger, C. (2001). "N-Carboxymethanofuran (carbamate) formation from methanofuran and CO2 in methanogenic archaea: Thermodynamics and kinetics of the spontaneous reaction". Eur. J. Biochem. 267 (11): 3130–3138. doi:10.1046/j.1432-1327.2000.01331.x. PMID 10824097.
- ^ T, Lundqvist; G, Schneider (1991-01-29). "Crystal Structure of the Ternary Complex of ribulose-1,5-bisphosphate Carboxylase, Mg(II), and Activator CO2 at 2.3-A Resolution". Biochemistry. 30 (4): 904–8. doi:10.1021/bi00218a004. PMID 1899197.
- ^ Adam, Norbert; Avar, Geza; Blankenheim, Herbert; Friederichs, Wolfgang; Giersig, Manfred; Weigand, Eckehard; Halfmann, Michael; Wittbecker, Friedrich-Wilhelm; Larimer (2005). "Polyurethanes". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a21_665.pub3. ISBN 978-3527306732.
- ^ Fukuto, T. R. (1990). "Mechanism of action of organophosphorus and carbamate insecticides". Environmental Health Perspectives. 87: 245–254. doi:10.1289/ehp.9087245. PMC 1567830. PMID 2176588.
- ^ a b "IRAC Mode of Action Classification Scheme Version 9.4" (pdf). Insecticide Resistance Action Committee. March 2020.
- ^ Metcalf, Robert L. "Insect Control". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a14_263. ISBN 978-3527306732.
- ^ "Pesticide Information Project: Fenoxycarb". Cornell University. Retrieved 15 June 2019.
- ^ National Center for Biotechnology Information. "PubChem Compound Summary for CID 125098, Icaridin". Pubchem. Retrieved 2024-01-08.
- ^ a b c Sparks, Thomas; Crossthwaite, Andrew; Nauen, Ralf; Banba, Shinichi; Cordova, Daniel; Earley, Fergus; Ebbinghaus-Kintscher, Ulrich; Fujioka, Shinsuke; Hirao, Ayako; Karmon, Danny; Kennedy, Robert; Nakao, Toshifumi; Popham, Holly; Salgado, Vincent; Watson, Gerald; Wedel, Barbara; Wessels, Frank (2020). "Insecticides, biologics and nematicides: Updates to IRAC's mode of action classification - a tool for resistance management". Pesticide Biochemistry and Physiology. 167. Elsevier: 104587. doi:10.1016/j.pestbp.2020.104587. ISSN 0048-3575. PMID 32527435.
- ^ Oakeshott, John; Devonshire, Alan; Claudianos, Charles; Sutherland, Tara; Horne, Irene; Campbell, Peter; Ollis, David; Russell, Robyn (2005). "Comparing the organophosphorus and carbamate insecticide resistance mutations in cholin- and carboxyl-esterases". Chemico-Biological Interactions. 157–158. Elsevier BV: 269–275. doi:10.1016/j.cbi.2005.10.041. ISSN 0009-2797. PMID 16289012. S2CID 32597626.
- ^ Gupta, Ramesh C, ed. (2015). Handbook of Toxicology of Chemical Warfare Agents. Cambridge, Massachusetts, United States: Academic Press. pp. 338–339. ISBN 978-0-12-800494-4.
- ^ Ellison, D (2008). Handbook of chemical and biological warfare agents. Boca Raton: CRC Press. ISBN 978-0-8493-1434-6. OCLC 82473582.
- ^ Badreshia, S. (2002). "Iodopropynyl butylcarbamate". Am. J. Contact Dermat. 13 (2): 77–79. doi:10.1053/ajcd.2002.30728. ISSN 1046-199X. PMID 12022126.
- ^ Vommina V. Sureshbabu; Narasimhamurthy Narendra (2011). "Protection Reactions". In Andrew B. Hughes (ed.). Protection Reactions, Medicinal Chemistry, Combinatorial Synthesis. Amino Acids, Peptides and Proteins in Organic Chemistry. Vol. 4. Wiley-VCH. pp. XVIII–LXXXIV. doi:10.1002/9783527631827.ch1. ISBN 9783527641574.
- ^ Wellings, Donald A.; Atherton, Eric (1997). "[4] Standard Fmoc protocols". Solid-Phase Peptide Synthesis. Methods in Enzymology. Vol. 289. pp. 44–67. doi:10.1016/s0076-6879(97)89043-x. ISBN 9780121821906. PMID 9353717.
- ^ Katsoyannis, P. G., ed. (1973). The Chemistry of Polypeptides. New York: Plenum Press. doi:10.1007/978-1-4613-4571-8. ISBN 978-1-4613-4571-8. S2CID 35144893. Archived from the original on 2022-10-13. Retrieved 2021-04-01.
- ^ Marullo, N. P.; Wagener, E. H. (1969-01-01). "Structural organic chemistry by nmr. III. Isomerization of compounds containing the carbon-nitrogen double bond". Tetrahedron Letters. 10 (30): 2555–2558. doi:10.1016/S0040-4039(01)88566-X.
- ^ Holland, J. R.; Hosley, H.; Scharlau, C.; Carbone, P. P.; Frei, E. III; Brindley, C. O.; Hall, T. C.; Shnider, B. I.; Gold, G. L.; Lasagna, L.; Owens, A. H. Jr; Miller, S. P. (1 March 1966). "A controlled trial of urethane treatment in multiple myeloma". Blood. 27 (3): 328–42. doi:10.1182/blood.V27.3.328.328. ISSN 0006-4971. PMID 5933438.
- ^ The Chemical/Biological Safety Section (CBSS) of the Office of Environmental Health and Safety. "Working with Urethane" (PDF). Virginia Commonwealth University. Archived from the original (PDF) on 2013-05-11.
- ^ Carbachol Multum Consumer Information. Accessed 27 April 2021.
- ^ DrugBank DB01264 . Accessed 27 April 2021.
- ^ Hikino H, Ogata K, Kasahara Y, Konno C (May 1985). "Pharmacology of ephedroxanes". Journal of Ethnopharmacology. 13 (2): 175–191. doi:10.1016/0378-8741(85)90005-4. PMID 4021515.
- ^ Colović, MB; Krstić, DZ; Lazarević-Pašti, TD; Bondžić, AM; Vasić, VM (2013). "Acetylcholinesterase inhibitors: pharmacology and toxicology". Curr Neuropharmacol. 11 (3): 315–35. doi:10.2174/1570159X11311030006. PMC 3648782. PMID 24179466.
- ^ Popovska-Gorevski, M; Dubocovich, ML; Rajnarayanan, RV (2017). "Carbamate Insecticides Target Human Melatonin Receptors". Chem Res Toxicol. 30 (2): 574–582. doi:10.1021/acs.chemrestox.6b00301. PMC 5318275. PMID 28027439.
- ^ a b c Marais, Simone; Diase, Elsa; Pereira, Maria de Lourdes (2012). "Carbamates: Human Exposure and Health Effects". The Impact of Pesticides. 21: 38 – via Research Gate.
- ^ Rüdiger Schubart (2000). "Dithiocarbamic Acid and Derivatives". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a09_001. ISBN 3-527-30673-0.
- ^ a b Walter, W.; Bode, K.-D. (April 1967). "Syntheses of Thiocarbamates". Angewandte Chemie International Edition in English. 6 (4): 281–293. doi:10.1002/anie.196702811.
- ^ Newman, Melvin S.; Hetzel, Frederick W. (1971). "Thiophenols from Phenols: 2-Naphthalenethiol". Org. Synth. 51: 139. doi:10.15227/orgsyn.051.0139.
- ^ A dictionary of chemistry. John Daintith (6th ed.). New York: Oxford University Press. 2008. ISBN 978-1-61583-965-0. OCLC 713875281.
{{cite book}}: CS1 maint: others (link) - ^ a b c Dumas, Jean (1833). "Recherches de chimie organique". Annales de Chimie et de Physique. 2nd series. 56: 496–556.
- ^ "urethane | Etymology, origin and meaning of urethane by etymonline". www.etymonline.com. Retrieved 2023-03-29.
- ^ a b Medlock, Henry (1849). "XXIX.—Researches on the amyl series". Q. J. Chem. Soc. 1 (4): 368–379. doi:10.1039/qj8490100368. ISSN 1743-6893.
- ^ "Definition of CARBAMATE". www.merriam-webster.com. Retrieved 2023-03-29.
- ^ Divers, Edward (1870-01-01). "XXVII.—On the precipitation of solutions of ammonium carbonate, sodium carbonate, and ammonium carbamate by calcium chloride". Journal of the Chemical Society. 23: 359–364. doi:10.1039/JS8702300359. ISSN 0368-1769.
- ^ "Carbamates (C00803)". Goldbook. IUPAC. doi:10.1351/goldbook.C00803. Retrieved 2023-03-29.
- ^ Moss, G. P.; Smith, P. a. S.; Tavernier, D. (1995-01-01). "Glossary of class names of organic compounds and reactivity intermediates based on structure (IUPAC Recommendations 1995)". Pure and Applied Chemistry (in German). 67 (8–9): 1307–1375. doi:10.1351/pac199567081307. ISSN 1365-3075. S2CID 95004254.


