
Summary
The hydrogen economy is an umbrella term that draws together the roles hydrogen can play alongside low-carbon electricity to decarbonize those sectors and activities which may be technically difficult to decarbonize through other means, or where cheaper and more energy-efficient clean solutions are not available.[2] In this context, hydrogen economy encompasses hydrogen's production through to end-uses in ways that substantively contribute to phasing-out fossil fuels and limiting climate change.
Most hydrogen produced today is gray hydrogen, made from natural gas through steam methane reforming (SMR) which accounted for 1.8% of global greenhouse gas emissions in 2021.[3] Low-carbon hydrogen, which is made using SMR with carbon capture and storage (blue hydrogen), or through electrolysis of water using renewable power (green hydrogen), accounted for under 1% of production.[4] Virtually all of the 100 million tonnes[5] of hydrogen produced each year is used in oil refining (43% in 2021) and industry (57%), principally in the manufacture of ammonia for fertilizers, and methanol.[6]: 18, 22, 29
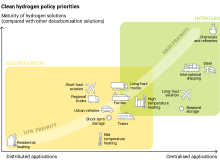
In its contribution to limiting global warming to 1.5 °C, it is broadly envisaged that the future hydrogen economy replaces gray hydrogen with blue and predominantly green hydrogen, produced in greater total volumes, to provide for an expanded set of end-uses.[7] These are likely to be in heavy industry (e.g. high temperature processes alongside electricity, feedstock for production of green ammonia and organic chemicals, as alternative to coal-derived coke for steelmaking), long-haul transport (e.g. shipping, aviation and to a lesser extent heavy goods vehicles), and long-term energy storage.[7][8] Other applications, such as light duty vehicles and heating in buildings, are no longer part of the future hydrogen economy, primarily for economic and environmental reasons.[9][10] These reasons include the difficulty of developing long-term storage, pipelines, and engine equipment, safety concerns since hydrogen is highly explosive, and the inefficiency of hydrogen compared to direct use of electricity.
As of 2023[update] there are no real alternatives to hydrogen for ammonia production for fertilizer, hydrogenation, hydrocracking, and hydrodesulfurization.[11] The extent to which hydrogen will be used to decarbonise chemical feedstock, long haul aviation and shipping, and long-term energy storage is likely to be influenced by the evolving production costs of low- and zero-carbon hydrogen. Estimates of future costs face numerous uncertainties – such as the introduction of carbon taxes, geography and geopolitics of energy, energy prices, technology choices, and their raw material requirements – but it is likely that green or white (underground[12]) hydrogen will see the greatest reductions in production cost over time.[13]
History and contemporary rationale edit
Origins edit
The concept of the hydrogen economy, though not the term, was by geneticist J.B.S. Haldane in 1923, who, anticipating the exhaustion of Britain's coal reserves for power generation, proposed a network of wind turbines to produce hydrogen for long-term energy storage through electrolysis, to help address renewable power's variable output.[14] The term itself was coined by John Bockris during a talk he gave in 1970 at General Motors (GM) Technical Center.[15] Bockris viewed it as an economy in which hydrogen, underpinned by nuclear and solar power, would help address growing concern about fossil fuel depletion and environmental pollution, by serving as energy carrier for end-uses in which electrification was not suitable.[2]
A hydrogen economy was proposed by the University of Michigan to solve some of the negative effects of using hydrocarbon fuels where the carbon is released to the atmosphere (as carbon dioxide, carbon monoxide, unburnt hydrocarbons, etc.). Modern interest in the hydrogen economy can generally be traced to a 1970 technical report by Lawrence W. Jones of the University of Michigan,[16] in which he echoed Bockris' dual rationale of addressing energy security and environmental challenges. Unlike Haldane and Bockris, Jones only focused on nuclear power as the energy source for electrolysis, and principally on the use of hydrogen in transport, where he regarded aviation and heavy goods transport as the top priorities.[17]
Later evolution edit
A spike in attention for the hydrogen economy concept during the 2000s was repeatedly described as hype by some critics and proponents of alternative technologies,[19][20][21] and investors lost money in the bubble.[22] Interest in the energy carrier resurged in the 2010s, notably with the forming of the World Hydrogen Council in 2017. Several manufacturers released hydrogen fuel cell cars commercially, with manufacturers such as Toyota, Hyundai, and industry groups in China having planned to increase numbers of the cars into the hundreds of thousands over the next decade.[23][24]
The global scope for hydrogen's role in cars is shrinking relative to earlier expectations.[25][26] By the end of 2022, 70,200 hydrogen vehicles had been sold worldwide,[27] compared with 26 million plug-in electric vehicles.[28]
Early 2020s takes on the hydrogen economy share earlier perspectives' emphasis on the complementarity of electricity and hydrogen, and the use of electrolysis as the mainstay of hydrogen production.[7] They focus on the need to limit global warming to 1.5 °C and prioritize the production, transportation and use of green hydrogen for heavy industry (e.g. high-temperature processes alongside electricity,[29] feedstock for production of green ammonia and organic chemicals,[7] as alternative to coal-derived coke for steelmaking),[30] long-haul transport (e.g. shipping, aviation and to a lesser extent heavy goods vehicles), and long-term energy storage.[7][8]
Current hydrogen market edit
Hydrogen production globally was valued at over US$155 billion in 2022 and is expected to grow over 9% annually through 2030.[31]
In 2021, 94 million tonnes (Mt) of molecular hydrogen (H2) was produced.[32] Of this total, approximately one sixth was as a by-product of petrochemical industry processes.[4] Most hydrogen comes from dedicated production facilities, over 99% of which is from fossil fuels, mainly via steam reforming of natural gas (70%) and coal gasification (30%, almost all of which in China).[4] Less than 1% of dedicated hydrogen production is low carbon: steam fossil fuel reforming with carbon capture and storage, green hydrogen produced using electrolysis, and hydrogen produced from biomass.[4] CO2 emissions from 2021 production, at 915 MtCO2,[33] amounted to 2.5% of energy-related CO2 emissions[34] and 1.8% of global greenhouse gas emissions.[3]
Virtually all hydrogen produced for the current market is used in oil refining (40 MtH2 in 2021) and industry (54 MtH2).[6]: 18, 22 In oil refining, hydrogen is used, in a process known as hydrocracking, to convert heavy petroleum sources into lighter fractions suitable for use as fuels. Industrial uses mainly comprise ammonia production to make fertilisers (34 MtH2 in 2021), methanol production (15 MtH2) and the manufacture of direct reduced iron (5 MtH2).[6]: 29
Uses edit
Hydrogen can be deployed as a fuel in two distinct ways: in fuel cells which produce electricity, and via combustion to generate heat.[36] When hydrogen is consumed in fuel cells, the only emission at the point of use is water vapour.[36] Combustion of hydrogen can lead to the thermal formation of harmful nitrogen oxides emissions.[36]
Industry edit
In the context of limiting global warming, low-carbon hydrogen (particularly green hydrogen) is likely to play an important role in decarbonising industry.[37] Hydrogen fuel can produce the intense heat required for industrial production of steel, cement, glass, and chemicals, thus contributing to the decarbonisation of industry alongside other technologies, such as electric arc furnaces for steelmaking.[29] However, it is likely to play a larger role in providing industrial feedstock for cleaner production of ammonia and organic chemicals.[37] For example, in steelmaking, hydrogen could function as a clean energy carrier and also as a low-carbon catalyst replacing coal-derived coke.[30]
The imperative to use low-carbon hydrogen to reduce greenhouse gas emissions has the potential to reshape the geography of industrial activities, as locations with appropriate hydrogen production potential in different regions will interact in new ways with logistics infrastructure, raw material availability, human and technological capital.[37]
Transport edit
Much of the interest in the hydrogen economy concept is focused on hydrogen vehicles, particularly planes.[38][39][40] Hydrogen vehicles produce significantly less local air pollution than conventional vehicles.[41] By 2050, the energy requirement for transportation might be between 20% and 30% fulfilled by hydrogen and synthetic fuels.[42][43][44]
Hydrogen used to decarbonise transportation is likely to find its largest applications in shipping, aviation and to a lesser extent heavy goods vehicles, through the use of hydrogen-derived synthetic fuels such as ammonia and methanol, and fuel cell technology.[7] Hydrogen has been used in fuel cell buses for many years. It is also used as a fuel for spacecraft propulsion.
In the International Energy Agency's 2022 Net Zero Emissions Scenario (NZE), hydrogen is forecast to account for 2% of rail energy demand in 2050, while 90% of rail travel is expected to be electrified by then (up from 45% today). Hydrogen's role in rail would likely be focused on lines that prove difficult or costly to electrify.[45] The NZE foresees hydrogen meeting approximately 30% of heavy truck energy demand in 2050, mainly for long-distance heavy freight (with battery electric power accounting for around 60%).[46]
Although hydrogen can be used in adapted internal combustion engines, fuel cells, being electrochemical, have an efficiency advantage over heat engines. Fuel cells are more expensive to produce than common internal combustion engines but also require higher purity hydrogen fuel than internal combustion engines.[47]
In the light road vehicle segment including passenger cars, by the end of 2022, 70,200 fuel cell electric vehicles had been sold worldwide,[27] compared with 26 million plug-in electric vehicles.[28] With the rapid rise of electric vehicles and associated battery technology and infrastructure, hydrogen's role in cars is minuscule.[25][26]
Energy system balancing and storage edit
Green hydrogen, from electrolysis of water, has the potential to address the variability of renewable energy output. Producing green hydrogen can both reduce the need for renewable power curtailment during periods of high renewables output and be stored long-term to provide for power generation during periods of low output.[48][49]
Ammonia edit
An alternative to gaseous hydrogen as an energy carrier is to bond it with nitrogen from the air to produce ammonia, which can be easily liquefied, transported, and used (directly or indirectly) as a clean and renewable fuel.[50][51] Among disadvantages of ammonia as an energy carrier are its high toxicity, energy efficiency of NH3 production from N2 and H2, and poisoning of PEM Fuel Cells by traces of non-decomposed NH3 after NH3 to N2 conversion.
Buildings edit
Numerous industry groups (gas networks, gas boiler manufacturers) across the natural gas supply chain are promoting hydrogen combustion boilers for space and water heating, and hydrogen appliances for cooking, to reduce energy-related CO2 emissions from residential and commercial buildings.[52][53][10] The proposition is that current end-users of piped natural gas can await the conversion of and supply of hydrogen to existing natural gas grids, and then swap heating and cooking appliances, and that there is no need for consumers to do anything now.[52][53][10]
A review of 32 studies on the question of hydrogen for heating buildings, independent of commercial interests, found that the economics and climate benefits of hydrogen for heating and cooking generally compare very poorly with the deployment of district heating networks, electrification of heating (principally through heat pumps) and cooking, the use of solar thermal, waste heat and the installation of energy efficiency measures to reduce energy demand for heat.[10] Due to inefficiencies in hydrogen production, using blue hydrogen to replace natural gas for heating could require three times as much methane, while using green hydrogen would need two to three times as much electricity as heat pumps.[10] Hybrid heat pumps, which combine the use of an electric heat pump with a hydrogen boiler, may play a role in residential heating in areas where upgrading networks to meet peak electrical demand would otherwise be costly.[10]
The widespread use of hydrogen for heating buildings would entail higher energy system costs, higher heating costs and higher environmental impacts than the alternatives, although a niche role may be appropriate in specific contexts and geographies.[10] If deployed, using hydrogen in buildings would drive up the cost of hydrogen for harder-to-decarbonise applications in industry and transport.[10]
Bio-SNG edit
As of 2019[update] although technically possible production of syngas from hydrogen and carbon-dioxide from bio-energy with carbon capture and storage (BECCS) via the Sabatier reaction is limited by the amount of sustainable bioenergy available:[54] therefore any bio-SNG made may be reserved for production of aviation biofuel.[55]
Hydrogen infrastructure edit
A hydrogen infrastructure is the infrastructure of hydrogen pipeline transport, points of hydrogen production and hydrogen stations (sometimes clustered as a hydrogen highway) for distribution as well as the sale of hydrogen fuel,[56] and thus a crucial prerequisite before a successful commercialization of automotive fuel cell technology.[57]
The hydrogen infrastructure would consist mainly of industrial hydrogen pipeline transport and hydrogen-equipped filling stations like those found on a hydrogen highway. Hydrogen stations which were not situated near a hydrogen pipeline would get supply via hydrogen tanks, compressed hydrogen tube trailers, liquid hydrogen trailers, liquid hydrogen tank trucks or dedicated onsite production.
Pipelines are the cheapest way to move hydrogen over long distances compared to other options. Hydrogen gas piping is routine in large oil-refineries, because hydrogen is used to hydrocrack fuels from crude oil.
Hydrogen embrittlement (a reduction in the ductility of a metal due to absorbed hydrogen) is not a problem for hydrogen gas pipelines. Hydrogen embrittlement only happens with 'diffusible' hydrogen, i.e. atoms or ions. Hydrogen gas, however, is molecular (H2), and there is a very significant energy barrier to splitting it into atoms.[58]
The IEA recommends existing industrial ports be used for production and existing natural gas pipelines for transport: also international co-operation and shipping.[59]
South Korea and Japan,[60] which as of 2019 lack international electrical interconnectors, are investing in the hydrogen economy.[61] In March 2020, the Fukushima Hydrogen Energy Research Field was opened in Japan, claiming to be the world's largest hydrogen production facility.[62] The site occupies 180,000 m2 (1,900,000 sq ft) of land, much of which is occupied by a solar array; power from the grid is also used for electrolysis of water to produce hydrogen fuel.[63]Production edit
Hydrogen gas is produced by several industrial methods. In 2022 less than 1% of hydrogen production was low-carbon.[64] Fossil fuels are the dominant source of hydrogen, for example by steam reforming of natural gas.[65] Other methods of hydrogen production include biomass gasification and methane pyrolysis. Methane pyrolysis and water electrolysis can use any source of electricity including renewable energy. Underground hydrogen is extracted.[66]
The production of hydrogen plays a key role in any industrialized society, since hydrogen is required for many chemical processes.[67] In 2020, roughly 87 million tons of hydrogen was produced[68] worldwide for various uses, such as oil refining, in the production of ammonia through the Haber process, and in the production of methanol through reduction of carbon monoxide. The global hydrogen generation market was fairly valued at US$155 billion in 2022, and expected to grow at a compound annual growth rate of 9.3% from 2023 to 2030.[69]
As of 2022[update], more than 95% of global hydrogen production is sourced from fossil gas and coal without carbon abatement.[70]: 1Storage edit
Several methods exist for storing hydrogen. These include mechanical approaches such as using high pressures and low temperatures, or employing chemical compounds that release H2 upon demand. While large amounts of hydrogen are produced by various industries, it is mostly consumed at the site of production, notably for the synthesis of ammonia. For many years hydrogen has been stored as compressed gas or cryogenic liquid, and transported as such in cylinders, tubes, and cryogenic tanks for use in industry or as propellant in space programs. The overarching challenge is the very low boiling point of H2: it boils around 20.268 K (−252.882 °C or −423.188 °F). Achieving such low temperatures requires expending significant energy.
Although molecular hydrogen has very high energy density on a mass basis, partly because of its low molecular weight, as a gas at ambient conditions it has very low energy density by volume. If it is to be used as fuel stored on board a vehicle, pure hydrogen gas must be stored in an energy-dense form to provide sufficient driving range. Because hydrogen is the smallest molecule, it easily escapes from containers. Considering leakages, transport and production costs, hydrogen could have a Global Warming Potential (GWP100) of 11.6. Methane, for comparison, has a GWP of 34.[71]Costs edit
More widespread use of hydrogen in economies entails the need for investment and costs in its production, storage, distribution and use. Estimates of hydrogen's cost are therefore complex and need to make assumptions about the cost of energy inputs (typically gas and electricity), production plant and method (e.g. green or blue hydrogen), technologies used (e.g. alkaline or proton exchange membrane electrolysers), storage and distribution methods, and how different cost elements might change over time.[72]: 49–65 These factors are incorporated into calculations of the levelized costs of hydrogen (LCOH). The following table shows a range of estimates of the levelized costs of gray, blue, and green hydrogen, expressed in terms of US$ per kg of H2 (where data provided in other currencies or units, the average exchange rate to US dollars in the given year are used, and 1 kg of H2 is assumed to have a calorific value of 33.3kWh).
| Production method | Note | Current cost (2020–2022) | Projected 2030 cost | Projected 2050 cost |
| Gray hydrogen (not including a carbon tax) | ||||
| International Energy Agency[73] | 2022 costs estimated for June, when gas prices peaked in the wake of Russia's invasion of Ukraine | 2021: 1.0–2.5 | – | – |
| 2022: 4.8–7.8 | ||||
| PWC[74] | 2021: 1.2–2.4 | |||
| Blue hydrogen | ||||
| International Energy Agency[73] | 2022 costs estimated for June, when gas prices peaked in the wake of Russia's invasion of Ukraine | 2021: 1.5–3.0 | – | – |
| 2022: 5.3–8.6 | ||||
| UK government[75] | Range dependent on gas price | 2020: 1.6–2.7 | 1.6–2.7 | 1.6–2.8 |
| GEP[76] | 2022: 2.8–3.5 | - | - | |
| Energy Transitions Commission[72]: 28 | 2020: 1.5–2.4 | 1.3–2.3 | 1.4–2.2 | |
| Green hydrogen | ||||
| International Energy Agency[73] | 2030 and 2050 estimates are using solar power in regions with good solar conditions | 2021: 4.0–9.0 | <1.5 | <1.0 |
| 2022: 3.0-4.3 | ||||
| UK government[75] | Using grid electricity, UK specific; range dependent on electricity price, and electrolyser technology and cost | 2020: 4.9–7.9 | 4.4–6.6 | 4.0–6.3 |
| Using otherwise curtailed renewable electricity, UK specific; range dependent on electrolyser technology and cost | 2020: 2.4–7.9 | 1.7–5.6 | 1.5–4.6 | |
| IRENA[77] | 2020: 2.2–5.2 | 1.4–4.1 | 1.1–3.4 | |
| GEP[76] | Source notes green H2 production cost has fallen by 60% since 2010 | 2022: 3.0–6.0 | ||
| Lazard[78] | 2022: 2.8–5.3 | |||
| PWC[74] | 2021: 3.5–9.5 | 1.8–4.8 | 1.2–2.4 | |
| Energy Transitions Commission[72]: 28 | 2020: 2.6–3.6 | 1.0–1.7 | 0.7–1.2 | |
The range of cost estimates for commercially available hydrogen production methods is broad, As of 2022, gray hydrogen is cheapest to produce without a tax on its CO2 emissions, followed by blue and green hydrogen. Blue hydrogen production costs are not anticipated to fall substantially by 2050,[75][72]: 28 can be expected to fluctuate with natural gas prices and could face carbon taxes for uncaptured emissions.[72]: 79 The cost of electrolysers fell by 60% from 2010 to 2022,[76] before rising slightly due to an increasing cost of capital.[22] Their cost is projected to fall significantly to 2030 and 2050,[79]: 26 driving down the cost of green hydrogen alongside the falling cost of renewable power generation.[80][72]: 28 It is cheapest to produce green hydrogen with surplus renewable power that would otherwise be curtailed, which favours electrolysers capable of responding to low and variable power levels.[79]: 5
A 2022 Goldman Sachs analysis anticipates that globally green hydrogen will achieve cost parity with grey hydrogen by 2030, earlier if a global carbon tax is placed on gray hydrogen.[13] In terms of cost per unit of energy, blue and gray hydrogen will always cost more than the fossil fuels used in its production, while green hydrogen will always cost more than the renewable electricity used to make it.
Subsidies for clean hydrogen production are much higher in the US and EU than in India.[81]
Examples and pilot programs edit
The distribution of hydrogen for the purpose of transportation is being tested around the world, particularly in the US (California, Massachusetts), Canada, Japan, the EU (Portugal, Norway, Denmark, Germany), and Iceland.
An indicator of the presence of large natural gas infrastructures already in place in countries and in use by citizens is the number of natural gas vehicles present in the country. The countries with the largest amount of natural gas vehicles are (in order of magnitude):[82] Iran, China, Pakistan, Argentina, India, Brasil, Italy, Colombia, Thailand, Uzbekistan, Bolivia, Armenia, Bangladesh, Egypt, Peru, Ukraine, United States. Natural gas vehicles can also be converted to run on hydrogen.
Also, in a few private homes, fuel cell micro-CHP plants can be found, which can operate on hydrogen, or other fuels as natural gas or LPG.[83][84]
Australia edit
Western Australia's Department of Planning and Infrastructure operated three Daimler Chrysler Citaro fuel cell buses as part of its Sustainable Transport Energy for Perth Fuel Cells Bus Trial in Perth.[85] The buses were operated by Path Transit on regular Transperth public bus routes. The trial began in September 2004 and concluded in September 2007. The buses' fuel cells used a proton exchange membrane system and were supplied with raw hydrogen from a BP refinery in Kwinana, south of Perth. The hydrogen was a byproduct of the refinery's industrial process. The buses were refueled at a station in the northern Perth suburb of Malaga.
In October 2021, Queensland Premier Annastacia Palaszczuk and Andrew Forrest announced that Queensland will be home to the world's largest hydrogen plant.[86]
In Australia, the Australian Renewable Energy Agency (ARENA) has invested $55 million in 28 hydrogen projects, from early stage research and development to early stage trials and deployments. The agency's stated goal is to produce hydrogen by electrolysis for $2 per kilogram, announced by Minister for Energy and Emissions Angus Taylor in a 2021 Low Emissions Technology Statement.[87]
European Union edit
Countries in the EU which have a relatively large natural gas pipeline system already in place include Belgium, Germany, France, and the Netherlands.[88] In 2020, The EU launched its European Clean Hydrogen Alliance (ECHA).[89][90]
France edit
Green hydrogen has become more common in France. A €150 million Green Hydrogen Plan was established in 2019, and it calls for building the infrastructure necessary to create, store, and distribute hydrogen as well as using the fuel to power local transportation systems like buses and trains. Corridor H2, a similar initiative, will create a network of hydrogen distribution facilities in Occitania along the route between the Mediterranean and the North Sea. The Corridor H2 project will get a €40 million loan from the EIB.[91][92]
Germany edit
German car manufacturer BMW has been working with hydrogen for years.[quantify].[93]
Iceland edit
Iceland has committed to becoming the world's first hydrogen economy by the year 2050.[94] Iceland is in a unique position. Presently,[when?] it imports all the petroleum products necessary to power its automobiles and fishing fleet. Iceland has large geothermal resources, so much that the local price of electricity actually is lower than the price of the hydrocarbons that could be used to produce that electricity.
Iceland already converts its surplus electricity into exportable goods and hydrocarbon replacements. In 2002, it produced 2,000 tons of hydrogen gas by electrolysis, primarily for the production of ammonia (NH3) for fertilizer. Ammonia is produced, transported, and used throughout the world, and 90% of the cost of ammonia is the cost of the energy to produce it.
Neither industry directly replaces hydrocarbons. Reykjavík, Iceland, had a small pilot fleet of city buses running on compressed hydrogen,[95] and research on powering the nation's fishing fleet with hydrogen is under way (for example by companies as Icelandic New Energy). For more practical purposes, Iceland might process imported oil with hydrogen to extend it, rather than to replace it altogether.
The Reykjavík buses are part of a larger program, HyFLEET:CUTE,[96] operating hydrogen fueled buses in eight European cities. HyFLEET:CUTE buses were also operated in Beijing, China and Perth, Australia (see below). A pilot project demonstrating a hydrogen economy is operational on the Norwegian island of Utsira. The installation combines wind power and hydrogen power. In periods when there is surplus wind energy, the excess power is used for generating hydrogen by electrolysis. The hydrogen is stored, and is available for power generation in periods when there is little wind.[citation needed]
India edit
India is said to adopt hydrogen and H-CNG, due to several reasons, amongst which the fact that a national rollout of natural gas networks is already taking place and natural gas is already a major vehicle fuel. In addition, India suffers from extreme air pollution in urban areas.[97][98] According to some estimates, nearly 80% of India's hydrogen is projected to be green, driven by cost declines and new production technologies.[99]
Currently however, hydrogen energy is just at the Research, Development and Demonstration (RD&D) stage.[100][101] As a result, the number of hydrogen stations may still be low,[102] although much more are expected to be introduced soon.[103][104][105]
Saudi Arabia edit
Saudi Arabia as a part of the NEOM project, is looking to produce roughly 1.2 million tonnes of green ammonia a year, beginning production in 2025.[106]
Turkey edit
The Turkish Ministry of Energy and Natural Resources and the United Nations Industrial Development Organization created the International Centre for Hydrogen Energy Technologies (UNIDO-ICHET) in Istanbul in 2004 and it ran to 2012.[107] In 2023 the ministry published a Hydrogen Technologies Strategy and Roadmap.[108]
United Kingdom edit
The UK started a fuel cell pilot program in January 2004, the program ran two Fuel cell buses on route 25 in London until December 2005, and switched to route RV1 until January 2007.[109] The Hydrogen Expedition is currently working to create a hydrogen fuel cell-powered ship and using it to circumnavigate the globe, as a way to demonstrate the capability of hydrogen fuel cells.[110] In August 2021 the UK Government claimed it was the first to have a Hydrogen Strategy and produced a document.[111]
In August 2021, Chris Jackson quit as chair of the UK Hydrogen and Fuel Cell Association, a leading hydrogen industry association, claiming that UK and Norwegian oil companies had intentionally inflated their cost projections for blue hydrogen in order to maximize future technology support payments by the UK government.[112]
United States edit
Several domestic U.S. automobile companies have developed vehicles using hydrogen, such as GM and Toyota.[113] However, as of February 2020, infrastructure for hydrogen was underdeveloped except in some parts of California.[114] The United States have their own hydrogen policy.[citation needed] A joint venture between NREL and Xcel Energy is combining wind power and hydrogen power in the same way in Colorado.[115] Hydro in Newfoundland and Labrador are converting the current wind-diesel Power System on the remote island of Ramea into a Wind-Hydrogen Hybrid Power Systems facility.[116]
A similar pilot project on Stuart Island uses solar power, instead of wind power, to generate electricity. When excess electricity is available after the batteries are fully charged, hydrogen is generated by electrolysis and stored for later production of electricity by fuel cell.[117] The US also have a large natural gas pipeline system already in place.[88]
See also edit
- Alternative fuel
- Combined cycle hydrogen power plant
- Energy development
- Hydrogen damage
- Hydrogen fuel cell power plant
- Hydrogen internal combustion engine vehicle
- Hydrogen prize
- Hydrogen-powered aircraft
- Hydrogen safety
- Lolland Hydrogen Community
- Methane pyrolysis
- Timeline of sustainable energy research 2020–present#Hydrogen energy
References edit
- ^ International Renewable Energy Agency (2022-03-29). "World Energy Transitions Outlook 1-5C Pathway 2022 edition". IRENA. p. 227. Retrieved 2023-10-06.
- ^ a b Yap, Jiazhen; McLellan, Benjamin (6 January 2023). "A Historical Analysis of Hydrogen Economy Research, Development, and Expectations, 1972 to 2020". Environments. 10 (1): 11. doi:10.3390/environments10010011. hdl:2433/284015. ISSN 2076-3298.
- ^ a b Greenhouse gas emissions totalled 49.3 Gigatonnes CO2e in 2021."Global Greenhouse Gas Emissions: 1990–2020 and Preliminary 2021 Estimates". Rhodium Group. 19 December 2022. Retrieved 2023-09-21.
- ^ a b c d "Hydrogen". IEA. 10 July 2023. "Energy" section. Retrieved 2023-09-21.
- ^ "Hydrogen". IEA. Retrieved 2024-03-24.
- ^ a b c IEA (2022). Global Hydrogen Review 2022. International Energy Agency. Retrieved 2023-08-25.
- ^ a b c d e f IPCC (2022). Shukla, P.R.; Skea, J.; Slade, R.; Al Khourdajie, A.; et al. (eds.). Climate Change 2022: Mitigation of Climate Change (PDF). Contribution of Working Group III to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge, UK and New York, NY, US: Cambridge University Press (In Press). pp. 91–92. doi:10.1017/9781009157926. ISBN 9781009157926.
- ^ a b IRENA (2021). "World Energy Transitions Outlook: 1.5 °C Pathway". International Renewable Energy Agency. Abu Dhabi. p. 95. Retrieved 2023-09-21.
- ^ Plötz, Patrick (2022-01-31). "Hydrogen technology is unlikely to play a major role in sustainable road transport". Nature Electronics. 5 (1): 8–10. doi:10.1038/s41928-021-00706-6. ISSN 2520-1131. S2CID 246465284.
- ^ a b c d e f g h Rosenow, Jan (September 2022). "Is heating homes with hydrogen all but a pipe dream? An evidence review". Joule. 6 (10): 2225–2228. doi:10.1016/j.joule.2022.08.015. S2CID 252584593.
- ^ Barnard, Michael (2023-10-22). "What's New On The Rungs Of Liebreich's Hydrogen Ladder?". CleanTechnica. Retrieved 2024-02-17.
- ^ Paddison, Laura (2023-10-29). "They went hunting for fossil fuels. What they found could help save the world". CNN. Retrieved 2024-02-17.
- ^ a b Goldman Sachs Research. "Carbonomics: The Clean Hydrogen Revolution". Goldman Sachs. pp. 4–6. Retrieved 2023-09-25.
- ^ "Daedalus or Science and the Future, A paper read to the Heretics, Cambridge, on February 4th, 1923 – Transcript 1993". Archived from the original on 2017-11-15. Retrieved 2016-01-16.
- ^ National Hydrogen Association; United States Department of Energy. "The History of Hydrogen" (PDF). hydrogenassociation.org. National Hydrogen Association. p. 1. Archived from the original (PDF) on 14 July 2010. Retrieved 17 December 2010.
- ^ Jones, Lawrence W (13 March 1970). Toward a liquid hydrogen fuel economy. University of Michigan Environmental Action for Survival Teach In. Ann Arbor, Michigan: University of Michigan. hdl:2027.42/5800.
- ^ Jones, Lawrence W. (March 13, 1970). Toward a Liquid Hydrogen Fuel Economy (PDF). pp. 2–3.
- ^ IRENA (2022), Geopolitics of the Energy Transformation: The Hydrogen Factor, International Renewable Energy Agency, Abu Dhabi. ISBN 978-92-9260-370-0.
- ^ Bakker, Sjoerd (2010). "The car industry and the blow-out of the hydrogen hype" (PDF). Energy Policy. 38 (11): 6540–6544. Bibcode:2010EnPol..38.6540B. doi:10.1016/j.enpol.2010.07.019. Archived (PDF) from the original on 2018-11-03. Retrieved 2019-12-11.
- ^ Harrison, James. "Reactions: Hydrogen hype". Chemical Engineer. 58: 774–775. Archived from the original on 2021-02-08. Retrieved 2017-08-31.
- ^ Rizzi, Francesco Annunziata, Eleonora Liberati, Guglielmo Frey, Marco (2014). "Technological trajectories in the automotive industry: are hydrogen technologies still a possibility?". Journal of Cleaner Production. 66: 328–336. doi:10.1016/j.jclepro.2013.11.069.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b "Can a viable industry emerge from the hydrogen shakeout?". The Economist. ISSN 0013-0613. Retrieved 2023-09-26.
- ^ Murai, Shusuke (2018-03-05). "Japan's top auto and energy firms tie up to promote development of hydrogen stations". The Japan Times Online. Japan Times. Archived from the original on 2018-04-17. Retrieved 16 April 2018.
- ^ Mishra, Ankit (2018-03-29). "Prospects of fuel-cell electric vehicles boosted with Chinese backing". Energy Post. Archived from the original on 2018-04-17. Retrieved 16 April 2018.
- ^ a b Plötz, Patrick (January 2022). "Hydrogen technology is unlikely to play a major role in sustainable road transport". Nature Electronics. 5 (1): 8–10. doi:10.1038/s41928-021-00706-6. ISSN 2520-1131. S2CID 246465284.
- ^ a b Collins (l_collins), Leigh (2022-02-02). "'Hydrogen unlikely to play major role in road transport, even for heavy trucks': Fraunhofer". Recharge | Latest Renewable Energy News. Retrieved 2023-09-08.
- ^ a b Chu, Yidan; Cui, Hongyang. Annual update on the global transition to electric vehicles: 2022 (PDF). International Council on Clean Transportation. pp. 2–3. Retrieved 2023-08-25.
- ^ a b Global EV Outlook 2023. IEA. 26 April 2023. pp. 14–24. Retrieved 2023-08-25.
- ^ a b Kjellberg-Motton, Brendan (2022-02-07). "Steel decarbonisation gathers speed | Argus Media". www.argusmedia.com. Retrieved 2023-09-07.
- ^ a b Blank, Thomas; Molly, Patrick (January 2020). "Hydrogen's Decarbonization Impact for Industry" (PDF). Rocky Mountain Institute. pp. 2, 7, 8. Archived (PDF) from the original on 22 September 2020.
- ^ "Hydrogen Generation Market Size, Share & Trends Analysis Report, 2023 – 2030". www.grandviewresearch.com. Retrieved 2023-08-30.
- ^ "Executive summary – Global Hydrogen Review 2022 – Analysis". IEA. Retrieved 2023-09-21.
- ^ "Hydrogen". IEA. Retrieved 2023-09-21.
- ^ Energy-related emissions totalled 36.3 Gigatonnes CO2 in 2021."Global CO2 emissions rebounded to their highest level in history in 2021 – News". IEA. Retrieved 2023-09-21.
- ^ Barnard, Michael (2023-10-22). "What's New On The Rungs Of Liebreich's Hydrogen Ladder?". CleanTechnica. Retrieved 2024-03-10.
- ^ a b c Lewis, Alastair C. (10 June 2021). "Optimising air quality co-benefits in a hydrogen economy: a case for hydrogen-specific standards for NO x emissions". Environmental Science: Atmospheres. 1 (5): 201–207. doi:10.1039/D1EA00037C. S2CID 236732702. This article incorporates text from this source, which is available under the CC BY 3.0 license.
- ^ a b c IPCC (2022). Shukla, P.R.; Skea, J.; Slade, R.; Al Khourdajie, A.; et al. (eds.). Climate Change 2022: Mitigation of Climate Change (PDF). Contribution of Working Group III to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge, UK and New York, NY, US: Cambridge University Press (In Press). p. 1184. doi:10.1017/9781009157926. ISBN 9781009157926.
- ^ "Is the time now ripe for planes to run on hydrogen?". The Economist. ISSN 0013-0613. Retrieved 2024-02-17.
- ^ Yusaf, Talal; Faisal Mahamude, Abu Shadate; Kadirgama, Kumaran; Ramasamy, Devarajan; Farhana, Kaniz; A. Dhahad, Hayder; Abu Talib, ABD Rahim (2024-01-02). "Sustainable hydrogen energy in aviation – A narrative review". International Journal of Hydrogen Energy. 52: 1026–1045. doi:10.1016/j.ijhydene.2023.02.086. ISSN 0360-3199.
- ^ Reid, Channing (2023-09-10). "H2FLY's Hydrogen-Powered Aircraft Has Completed Its First Flight". Simple Flying. Retrieved 2024-02-17.
- ^ "This company may have solved one of the hardest problems in clean energy". Vox. 2018-02-16. Archived from the original on 2019-11-12. Retrieved 9 February 2019.
- ^ IRENA. "The Hydrogen Factor". irena.org. Retrieved 2022-10-19.
- ^ "Sustainable fuels and their role in decarbonizing energy | McKinsey". www.mckinsey.com. Retrieved 2022-10-19.
- ^ Spiryagin, Maksym; Dixon, Roger; Oldknow, Kevin; Cole, Colin (2021-09-01). "Preface to special issue on hybrid and hydrogen technologies for railway operations". Railway Engineering Science. 29 (3): 211. doi:10.1007/s40534-021-00254-x. ISSN 2662-4753. S2CID 240522190.
- ^ World energy outlook 2022. International Energy Agency. p. 150. This article incorporates text from this source, which is available under the CC BY 4.0 license.
- ^ Cozzi, Laura; Gould, Tim. World Energy Outlook 2022 (PDF). International Energy Agency. p. 148. This article incorporates text from this source, which is available under the CC BY 4.0 license.
- ^ Stępień, Zbigniew (January 2021). "A Comprehensive Overview of Hydrogen-Fueled Internal Combustion Engines: Achievements and Future Challenges". Energies. 14 (20): 6504. doi:10.3390/en14206504. ISSN 1996-1073.
- ^ Schrotenboer, Albert H.; Veenstra, Arjen A.T.; uit het Broek, Michiel A.J.; Ursavas, Evrim (October 2022). "A Green Hydrogen Energy System: Optimal control strategies for integrated hydrogen storage and power generation with wind energy" (PDF). Renewable and Sustainable Energy Reviews. 168: 112744. doi:10.1016/j.rser.2022.112744. S2CID 250941369.
- ^ Lipták, Béla (January 24, 2022). "Hydrogen is key to sustainable green energy". Control. Retrieved February 12, 2023.
- ^ Agosta, Vito (July 10, 2003). "The Ammonia Economy". Archived from the original on May 13, 2008. Retrieved 2008-05-09.
- ^ "Renewable Energy". Iowa Energy Center. Archived from the original on 2008-05-13. Retrieved 2008-05-09.
- ^ a b Collins, Leigh (2021-12-10). "Even the European gas lobby can't make a case for hydrogen boilers — so why does it say gases are needed to decarbonise heating?". Recharge | Latest renewable energy news. Retrieved 2023-09-25.
- ^ a b Roth, Sammy (2023-02-09). "California declared war on natural gas. Now the fight is going national". Los Angeles Times. Retrieved 2023-09-25.
- ^ UKCCC H2 2018, p. 79: The potential for bio-gasification with CCS to be deployed at scale is limited by the amount of sustainable bioenergy available. .... "
- ^ UKCCC H2 2018, p. 33: production of biofuels, even with CCS, is only one of the best uses of the finite sustainable bio-resource if the fossil fuels it displaces cannot otherwise feasibly be displaced (e.g. use of biomass to produce aviation biofuels with CCS)."
- ^ "Hydrogen infrastructure project launches in USA". 14 May 2013.
- ^ Eberle, Ulrich; Mueller, Bernd; von Helmolt, Rittmar. "Fuel cell electric vehicles and hydrogen infrastructure: status 2012". Energy & Environmental Science. Retrieved 23 December 2014.
- ^ Bhadhesia, Harry. "Prevention of Hydrogen Embrittlement in Steels" (PDF). Phase Transformations & Complex Properties Research Group, Cambridge University. Archived (PDF) from the original on 11 November 2020. Retrieved 17 December 2020.
- ^ IEA H2 2019, p. 15
- ^ "Japan's Hydrogen Strategy and Its Economic and Geopolitical Implications". Etudes de l'Ifri. Archived from the original on 10 February 2019. Retrieved 9 February 2019.
- ^ "South Korea's Hydrogen Economy Ambitions". The Diplomat. Archived from the original on 9 February 2019. Retrieved 9 February 2019.
- ^ "The world's largest-class hydrogen production, Fukushima Hydrogen Energy Research Field (FH2R) now is completed at Namie town in Fukushima". Toshiba Energy Press Releases. Toshiba Energy Systems and Solutions Corporations. 7 March 2020. Archived from the original on 22 April 2020. Retrieved 1 April 2020.
- ^ Patel, Sonal (2022-07-01). "Fukushima Hydrogen Energy Research Field Demonstrates Hydrogen Integration". POWER Magazine. Retrieved 2023-10-05.
- ^ "Hydrogen". IEA. Retrieved 2024-03-23.
- ^ Häussinger, Peter; Lohmüller, Reiner; Watson, Allan M. (2011). "Hydrogen, 1. Properties and Occurrence". Ullmann's Encyclopedia of Industrial Chemistry. doi:10.1002/14356007.a13_297.pub2. ISBN 978-3-527-30673-2.
- ^ "Natural Hydrogen: A Potential Clean Energy Source Beneath Our Feet". Yale E360. Retrieved 2024-03-23.
- ^ Energy, U. S. D. o. The Impact of Increased Use of Hydrogen on Petroleum Consumption and Carbon Dioxide Emissions. 84 (Energy Information Administration, Washington, DC, 2008)
- ^ Collins, Leigh (2021-05-18). "A net-zero world 'would require 306 million tonnes of green hydrogen per year by 2050': IEA | Recharge". Recharge | Latest renewable energy news. Archived from the original on 2021-05-21.
- ^ "Global Hydrogen Generation Market Size Report, 2030".
- ^ Rosenow, Jan (27 September 2022). "Is heating homes with hydrogen all but a pipe dream? An evidence review". Joule. 6 (10): 2225–2228. doi:10.1016/j.joule.2022.08.015. ISSN 2542-4785. S2CID 252584593. Article in press.
- ^ Sand, Maria; Skeie, Ragnhild Bieltvedt; Sandstad, Marit; Krishnan, Srinath; Myhre, Gunnar; Bryant, Hannah; Derwent, Richard; Hauglustaine, Didier; Paulot, Fabien; Prather, Michael; Stevenson, David (2023). "A multi-model assessment of the Global Warming Potential of hydrogen". Communications Earth & Environment. 4: 203. doi:10.1038/s43247-023-00857-8. Retrieved 2024-04-11.
- ^ a b c d e f Making the Hydrogen Economy Possible: Accelerating Clean Hydrogen in an Electrified Economy. Energy Transitions Commission. April 2021. Retrieved 2023-08-25.
- ^ a b c Global Hydrogen Review 2022. IEA. p. 93. Retrieved 2023-08-25.
- ^ a b PricewaterhouseCoopers. "Green hydrogen economy – predicted development of tomorrow". PwC. Retrieved 2023-08-25.
- ^ a b c "Hydrogen Production Costs 2021 annex: Key assumptions and outputs for production technologies". GOV.UK. Retrieved 2023-08-25.
- ^ a b c Saini, Anshuman (January 12, 2023). "Green & Blue Hydrogen: Current Levelized Cost of Production & Outlook | GEP Blogs". www.gep.com. Retrieved 2023-08-25.
- ^ IRENA (2020), Green Hydrogen Cost Reduction: Scaling up Electrolysers to Meet the 1.5 °C Climate Goal, International Renewable Energy Agency, Abu Dhabi, p. 91.
- ^ 2023 Levelized Cost Of Energy+. Lazard. April 12, 2023. p. 27. Retrieved 2023-08-25.
- ^ a b Patonia, Aliaksei; Poudineh, Rahmat (January 2022). Cost-competitive green hydrogen: how to lower the cost of electrolysers?. Oxford Institute for Energy Studies. Retrieved 2023-08-25.
- ^ Roser, Max (2023-09-01). "Why did renewables become so cheap so fast?". Our World in Data.
- ^ Martin, Polly (2023-06-29). "India to offer green hydrogen production subsidy of up to $0.60/kg — for three years only". Hydrogen news and intelligence | Hydrogen Insight. Retrieved 2023-09-26.
- ^ "Worldwide NGV statistics". Archived from the original on 2015-02-06. Retrieved 2019-09-29.
- ^ "Fuel Cell micro CHP". Archived from the original on 2019-11-06. Retrieved 2019-10-23.
- ^ "Fuel cell micro Cogeneration". Archived from the original on 2019-10-23. Retrieved 2019-10-23.
- ^ "Perth Fuel Cell Bus Trial". Department for Planning and Infrastructure, Government of Western Australia. 13 April 2007. Archived from the original on 7 June 2008. Retrieved 2008-05-09.
- ^ "'Green industrial revolution': Queensland announces plans to mass produce green ammonia". ABC News. October 11, 2021. Archived from the original on 2021-10-12. Retrieved 2021-10-12 – via www.abc.net.au.
- ^ "Australia's pathway to $2 per kg hydrogen – ARENAWIRE". Australian Renewable Energy Agency. 30 November 2020. Archived from the original on 2020-12-15. Retrieved 2021-01-06.
- ^ a b "Hydrogen transport & distribution". Archived from the original on 2019-09-29. Retrieved 2019-09-29.
- ^ Pollet, Mathieu (2020). "AExplainer: Why is the EU Commission betting on hydrogen for a greener future?". euronews. Archived from the original on 2020-08-07. Retrieved 2020-08-14.
- ^ "ECHA". Archived from the original on 2020-08-12. Retrieved 2020-08-14.
- ^ "French port bets big on floating wind farms planned in Mediterranean". European Investment Bank. Retrieved 2022-09-26.
- ^ "Green Hydrogen: A key investment for the energy transition". blogs.worldbank.org. 23 June 2022. Retrieved 2022-09-26.
- ^ "E3B1C256-BFCB-4CEF-88A6-1DCCD7666635". 24 October 2007. Archived from the original on 2021-10-29. Retrieved 2021-10-12.
- ^ Hannesson, Hjálmar W. (2007-08-02). "Climate change as a global challenge". Iceland Ministry for Foreign Affairs. Archived from the original on 2014-01-07. Retrieved 2008-05-09.
- ^ Doyle, Alister (January 14, 2005). "Iceland's hydrogen buses zip toward oil-free economy". Reuters. Archived from the original on July 24, 2012. Retrieved 2008-05-09.
- ^ "What is HyFLEET:CUTE?". Archived from the original on 2008-02-24. Retrieved 2008-05-09.
- ^ "Hydrogen vehicles and refueling infrastructure in India" (PDF). Archived (PDF) from the original on 2017-06-12. Retrieved 2019-09-28.
- ^ Das, L (1991). "Exhaust emission characterization of hydrogen-operated engine system: Nature of pollutants and their control techniques". International Journal of Hydrogen Energy. 16 (11): 765–775. doi:10.1016/0360-3199(91)90075-T.
- ^ "UK-India Energy Collaborations report" (PDF).
- ^ "MNRE: FAQ". Archived from the original on 2019-09-21. Retrieved 2019-09-28.
- ^ Overview of Indian Hydrogen Programme
- ^ "H2 stations worldwide". Archived from the original on 2019-09-21. Retrieved 2019-09-28.
- ^ "India working on more H2 stations". 23 February 2016. Archived from the original on 2019-09-21. Retrieved 2019-09-28.
- ^ "Shell plans to open 1200 fuel stations in India, some of which may include H2 refilling". The Economic Times. Archived from the original on 2019-09-22. Retrieved 2019-09-28.
- ^ "Hydrogen Vehicles and Refueling Infrastructure in India" (PDF). Archived (PDF) from the original on 2017-06-12. Retrieved 2019-09-28.
- ^ "Saudi Arabia's $5bn green hydrogen-based ammonia plant to begin production in 2025". Energy & Utilities. 21 April 2021. Retrieved 2022-01-13.
- ^ "Independent Mid-Term Review of the UNIDO Project: Establishment and operation of the International Centre for Hydrogen Energy Technologies (ICHET), TF/INT/03/002" (PDF). UNIDO. 31 August 2009. Archived from the original (PDF) on 1 June 2010. Retrieved 20 July 2010.
- ^ "Announcement – Republic of Türkiye Ministry of Energy and Natural Resources". enerji.gov.tr. Retrieved 2024-02-14.
- ^ "Hydrogen buses". Transport for London. Archived from the original on March 23, 2008. Retrieved 2008-05-09.
- ^ "The Hydrogen Expedition" (PDF). January 2005. Archived from the original (PDF) on 2008-05-27. Retrieved 2008-05-09.
- ^ "UK Hydrogen Strategy" (PDF). UK Government. August 2021. Archived (PDF) from the original on 2021-08-19. Retrieved 2021-08-19.
- ^ Ambrose, Jillian (20 August 2021). "Oil firms made 'false claims' on blue hydrogen costs, says ex-lobby boss". The Guardian. London, United Kingdom. ISSN 0261-3077. Archived from the original on 2021-08-24. Retrieved 2021-08-24.
- ^ "Are hydrogen fuel cell vehicles the future of autos?". ABC News. Archived from the original on 2021-01-17. Retrieved 2021-01-18.
- ^ Siddiqui, Faiz. "The plug-in electric car is having its moment. But despite false starts, Toyota is still trying to make the fuel cell happen". Washington Post. ISSN 0190-8286. Archived from the original on 2021-01-19. Retrieved 2021-01-18.
- ^ "Experimental 'wind to hydrogen' system up and running". Physorg.com. January 8, 2007. Archived from the original on 2013-01-26. Retrieved 2008-05-09.
- ^ "Hydrogen Engine Center Receives Order for Hydrogen Power Generator 250kW Generator for Wind/Hydrogen Demonstration" (PDF). Hydrogen Engine Center, Inc. May 16, 2006. Archived from the original (PDF) on May 27, 2008. Retrieved 2008-05-09.
- ^ "Stuart Island Energy Initiative". Archived from the original on 2013-06-18. Retrieved 2008-05-09.
Sources edit
- Hydrogen in a low-carbon economy (PDF). UK Committee on Climate Change. 2018.
- The Future of Hydrogen. International Energy Agency. 2019.
External links edit
- International Partnership for the Hydrogen Economy
- Hydrogen. International Energy Agency. 2022
- European Hydrogen Association
- Online calculator for green hydrogen production and transport costs


