
Summary
Mercury(II) cyanide, also known as mercuric cyanide, is a poisonous compound of mercury and cyanide. It is an odorless, toxic white powder. It is highly soluble in polar solvents such as water, alcohol, and ammonia; slightly soluble in ether; and insoluble in benzene and other hydrophobic solvents.[3]
| |
| Names | |
|---|---|
| IUPAC name
dicyanomercury
| |
| Other names
mercuric cyanide; cyanomercury; neutral mercury cyanide (1:2); mercury dicyanide; hydrargyri cyanidum[1] (homeopathy)
| |
| Identifiers | |
| |
3D model (JSmol)
|
|
| 3679510 | |
| ChEBI |
|
| ChemSpider |
|
| ECHA InfoCard | 100.008.857 |
| EC Number |
|
| 2563 | |
PubChem CID
|
|
| UNII |
|
| UN number | 1636 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| Hg(CN)2 | |
| Molar mass | 252.63 g/mol |
| Appearance | colorless crystals or white powder |
| Odor | odorless |
| Density | 3.996 g/cm3 |
| Melting point | 320 °C (608 °F; 593 K)[3] (decomposes) |
| 9.3 g/100 mL (14 °C) 53.9 g/100 mL (100 °C)[2] | |
| Solubility | 25 g/100 mL (methanol, 19.5 °C) soluble in ethanol, ammonia, glycerin slightly soluble in ether insoluble in benzene |
| −67.0·10−6 cm3/mol | |
Refractive index (nD)
|
1.645 |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Highly toxic |
| GHS labelling:[4] | |
  
| |
| Danger | |
| H300, H301, H310, H330, H373, H410 | |
| P260, P262, P264, P270, P271, P273, P280, P284, P301+P310, P302+P350, P304+P340, P310, P314, P320, P322, P330, P361, P363, P391, P403+P233, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
26 mg/kg |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Molecular and crystal structure edit
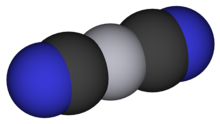
At ambient temperature and ambient pressure, Hg(CN)2 takes the form of tetragonal crystals.[3] These crystals are composed of nearly linear Hg(CN)2 molecules with a C-Hg-C bond angle of 175.0° and an Hg-C-N bond angle of 177.0° (Aylett[2] gives slightly different values of 189° and 175°, respectively). Raman spectra show that the molecules distort at higher pressures. Between 16-20 kbar, the structure undergoes a phase transition as the Hg(II) center changes from 2- to 4-coordinate as the CN groups bind to neighboring Hg centers forming via Hg-N bonds. The coordination geometry thus changes from tetragonal to tetrahedral, forming a cubic crystal structure, analogous to the structure of Cd(CN)2. Due to the ambidentate nature of the CN ligands, this tetrahedral structure is distorted, but the distortion lessens with increasing pressure until the structure becomes nearly perfectly tetrahedral at >40 kbar.[6]
As in the solid state, in aqueous solution, Hg(CN)2 molecules are linear.[2]
Synthesis edit
Mercuric cyanide can be prepared by mixing yellow mercury oxide with hydrocyanic acid in the following chemical reaction[2] which is generally carried out by passing HCN gas into HgO in water. When soluble Hg(CN)2 is formed, the solution is evaporated to crystallize the product.[1]
- HgO + 2 HCN → Hg(CN)2 + H2O
Hg(CN)2 can also be prepared by mixing HgO with finely powdered Prussian blue.[2][7] In addition, it can be produced by reacting mercuric sulfate with potassium ferrocyanide in water:[7]
- K4Fe(CN)6 + 3 HgSO4 → 3 Hg(CN)2 + 2 K2SO4 + FeSO4
Another method to generate mercuric cyanide is through the disproportionation of mercury(I) derivatives. In these reactions, metallic mercury precipitates, and Hg(CN)2 remains in solution:[7]
- Hg2(NO3)2 + 2 KCN → Hg + Hg(CN)2 + 2 KNO3
Reactions edit
It rapidly decomposes in acid to give off hydrogen cyanide. It is photosensitive, becoming darker in color.[8]
Mercury cyanide catalyzes the Koenigs–Knorr reaction for the synthesis of glycosides.[3] Cyanogen, (CN)2, forms upon heating dry mercury cyanide, but the method is inferior to other routes:[9]
- Hg(CN)2 → (CN)2 + Hg
Coordination polymers can be synthesized from Hg(CN)2 building blocks. Large single crystals of [(tmeda)Cu-[Hg(CN)2]2][HgCl4] form upon treating CuCl2, the soft Lewis acid Hg(CN)2, and N,N,N',N'-tetramethylethylenediamine (TMEDA). The migration of two labile chloride ligands from harder Cu(II) to softer Hg(II) drives the formation of the crystal.[10]
Past applications edit
The use of mercuric cyanide as an antiseptic was discontinued due to its toxicity.[11] Hg(CN)2 is also used in photography.[12]
Toxicology edit
Mercury(II) cyanide is poison with health hazard classification 3, having an oral LD50 of 33 milligrams per kilogram in mice and a subcutaneous LD50 of 2.7 milligrams per kilogram in dogs.[13]
References edit
- ^ a b "Hydrargyrum. Mercury. Part 5." http://chestofbooks.com/health/materia-medica-drugs/Manual-Pharmacology/Hydrargyrum-Mercury-Part-5.html (accessed April 1, 2009).
- ^ a b c d e Aylett, B.J. "Mercury (II) Pseudohalides: Cyanide, Thiocyanate, Selenocyanate, Azide, Fulminate." Comprehensive Inorganic Chemistry 3:304-306. J.C. Bailar, Harry Julius Emeléus, Sir Ronald Nyholm, and A.F. Trotman-Dickenson, ed. Oxford: Pergamon Press, 1973; distributed by Compendium Publishers (Elmsford, NY), p. 304.
- ^ a b c d Kocovsky, P., G. Wang, and V. Sharma. "Mercury(II) Cyanide." e-EROS Encyclopedia of Reagents for Organic Synthesis. Chichester, UK: John Wiley & Sons, Ltd., 2001. http://www.mrw.interscience.wiley.com/eros/articles/rm034/sect0-fs.html
- ^ "Mercuric cyanide". pubchem.ncbi.nlm.nih.gov. Retrieved 23 December 2021.
- ^ "MERCURIC CYANIDE | CAMEO Chemicals | NOAA".
- ^ Wong, P.T.T. J. Chem. Phys. 1984, 80(12), 5937-41.
- ^ a b c Miller, W.L. Elements of Chemistry: Organic chemistry, 5th ed. New York: John Wiley & Sons, 1880, p. 100.
- ^ Brunton, L.T. A Text-Book Of Pharmacology, Therapeutics And Materia Medica. London: MacMillan & Co., 1885.
- ^ Brotherton, T.K.; Lynn, J.W. Chemical Reviews 1959, 59(5), 841-883, 844-846.
- ^ Draper, Neil D.; Batchelor, Raymond J.; Sih, Bryan C.; Ye, Zuo-Guang; Leznoff, Daniel B. (2003). "Synthesis, Structure, and Properties of [(tmeda)Cu[Hg(CN)2]2][HgCl4]: A Non-Centrosymmetric 2-D Layered System that Shows Strong Optical Anisotropy". Chemistry of Materials. 15 (8): 1612–1616. doi:10.1021/cm021716r.
- ^ Benaissa, M.L.; Hantson, P.; Bismuth, C.; Baud, F.J. Intensive Care Med. 1995, 21(12), 1051-1053.
- ^ "Cyanides, Cyanide Oxides and Complex Cyanides." http://www.dncustoms.gov.vn/web_eglish/bieu_thue/E_HTM/E2837.HTM Archived 2017-12-30 at the Wayback Machine (accessed April 30, 2009).
- ^ Pubchem. "Mercuric cyanide". pubchem.ncbi.nlm.nih.gov. Retrieved 2018-03-22.
External links edit
- National Pollutant Inventory: Cyanide compounds fact sheet
- National Pollutant Inventory: Mercury and compounds fact sheet



