
Summary
Octaoxygen, also known as ε-oxygen or red oxygen, is an allotrope of oxygen consisting of eight oxygen atoms. This allotrope forms above 600 K (327 °C; 620 °F) at pressures greater than 17 GPa.[1]
 |
 |
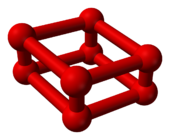
Ball-and-stick model of O8 |
Part of the crystal structure of ε-oxygen
|
Preparation and properties edit
As the pressure of oxygen at room temperature is increased through 10 gigapascals (1,500,000 psi), it undergoes a dramatic phase transition to a different allotrope. Its volume decreases significantly,[2] and it changes color from sky-blue to deep red.[3] This ε-phase was discovered in 1979, but the structure has been unclear. Based on infrared spectroscopy, researchers assumed in 1999 that this phase consists of O
4 molecules in a crystal lattice.[4] However, in 2006, it was shown by X-ray crystallography that this stable phase is in fact O
8.[5][6] No one predicted the structure theoretically:[1] a rhomboid O
8 cluster[7] consisting of four O
2 molecules.
In this phase, it exhibits a dark-red color, very strong infrared absorption, and a magnetic collapse.[8] It is also stable over a very large pressure domain[citation needed] and has been the subject of numerous X-ray diffraction, spectroscopic and theoretical studies. It has been shown to have a monoclinic C2/m symmetry, and its infrared absorption behaviour was attributed to the association of oxygen molecules into larger units. At 11 GPa, the intra-cluster bond length of the O
8 cluster is 0.234 nm, and the inter-cluster distance is 0.266 nm, both longer than the 0.120 nm bond-length in the oxygen molecule O
2.[1]
The formation mechanism of the O
8 cluster found in the work is not clear yet, and the researchers think that the charge transfer between oxygen molecules or the magnetic moment of oxygen molecules has a significant role in the formation.[1]
Potential applications edit
Liquid oxygen is already used as an oxidant in rockets, and it has been speculated that octaoxygen could make an even better oxidant, because of its higher energy density.[9]
Researchers think that this structure may greatly influence the structural investigation of elements.[1]
References edit
- ^ a b c d e Advanced Industrial Science and Technology (AIST) (2006). "Solid Oxygen ε-Phase Crystal Structure Determined Along With The Discovery of a Red Oxygen O8 Cluster". AZoNano. Retrieved 2008-01-10.
- ^ Akahama, Yuichi; Kawamura, Haruki; Häusermann, Daniel; Hanfland, Michael; Shimomura, Osamu (June 1995). "New High-Pressure Structural Transition of Oxygen at 96 GPa Associated with Metallization in a Molecular Solid". Physical Review Letters. 74 (23): 4690–4694. Bibcode:1995PhRvL..74.4690A. doi:10.1103/PhysRevLett.74.4690. PMID 10058574.
- ^ Nicol, Malcolm; Hirsch, K. R.; Holzapfel, Wilfried B. (December 1979). "Oxygen Phase Equilibria near 298 K". Chemical Physics Letters. 68 (1): 49–52. Bibcode:1979CPL....68...49N. doi:10.1016/0009-2614(79)80066-4.
- ^ Gorelli, Federico A.; Ulivi, Lorenzo; Santoro, Mario; Bini, Roberto (November 1999). "The ε Phase of Solid Oxygen: Evidence of an O4 Molecule Lattice". Physical Review Letters. 83 (20): 4093–4096. Bibcode:1999PhRvL..83.4093G. doi:10.1103/PhysRevLett.83.4093.
- ^ Fujihisa, Hiroshi; Akahama, Yuichi; Kawamura, Haruki; Ohishi, Yasuo; Shimomura, Osamu; Yamawaki, Hiroshi; Sakashita, Mami; Gotoh, Yoshito; Takeya, Satoshi; onda, Kazumasa H (2006-08-26). "O8 Cluster Structure of the Epsilon Phase of Solid Oxygen". Physical Review Letters. 97 (8): 085503. Bibcode:2006PhRvL..97h5503F. doi:10.1103/PhysRevLett.97.085503. PMID 17026315.
- ^ Lundegaard, Lars F.; Weck, Gunnar; McMahon, Malcolm I.; Desgreniers, Serge; Loubeyre, Paul (2006-09-14). "Observation of an O8 molecular lattice in the phase of solid oxygen". Nature. 443 (7108): 201–204. Bibcode:2006Natur.443..201L. doi:10.1038/nature05174. PMID 16971946. S2CID 4384225.
- ^ Steudel, Ralf; Wong, Ming Wah (2007). "Dark-Red O8 Molecules in Solid Oxygen: Rhomboid Clusters, Not S8-Like Rings". Angewandte Chemie International Edition. 46 (11): 1768–1771. doi:10.1002/anie.200604410. PMID 17450606.
- ^ Freiman, Yu A.; Jodl, H. J. (2004). "Solid oxygen". Physics Reports. 401 (1–4): 1–228. Bibcode:2004PhR...401....1F. doi:10.1016/j.physrep.2004.06.002.
- ^ Ball, Phillip (16 November 2001). "New form of oxygen found". Nature News. doi:10.1038/news011122-3.


