Summary
Parkinson's disease (PD), or simply Parkinson's, is a chronic degenerative disorder of the central nervous system that affects both the motor system and non-motor systems. The symptoms usually emerge slowly, and as the disease progresses, non-motor symptoms become more common. Early symptoms are tremor, rigidity, slowness of movement, and difficulty with walking. Problems may also arise with cognition, behaviour, sleep, and sensory systems. Parkinson's disease dementia is common in advanced stages.
| Parkinson's disease | |
|---|---|
| Other names | Parkinson disease, idiopathic or primary parkinsonism, hypokinetic rigid syndrome, paralysis agitans, shaking palsy |
A. 1880s illustration of Parkinson's disease (PD) B. Mild motor-predominant PD C. Intermediate PD D. Diffuse malignant PD | |
| Specialty | Neurology |
| Symptoms | Rigidity, slowness of movement, tremor, difficulty walking[1] |
| Complications | Dementia, depression, anxiety,[2] eating problems, and sleep problems |
| Usual onset | Age over 60[1][3] |
| Causes | Unknown[4] |
| Risk factors | Pesticide exposure, head injuries[4] |
| Diagnostic method | Based on symptoms[1] |
| Differential diagnosis | Dementia with Lewy bodies, progressive supranuclear palsy, essential tremor, antipsychotic use[5] |
| Treatment | Medications, surgery[1] |
| Medication | L-DOPA, dopamine agonists[2] |
| Frequency | 6.2 million (2015)[6] |
| Deaths | 117,400 (2015)[7] |
| Named after | James Parkinson |
The motor symptoms of the disease result from the nerve cell death in the substantia nigra, a midbrain region that supplies dopamine to the basal ganglia. The cause of this cell death is poorly understood but involves the aggregation of the protein alpha-synuclein into Lewy bodies within the neurons. Collectively, the main motor symptoms are known as parkinsonism. Contributing factors include a combination of genetic and environmental factors. Those with an affected family member are at an increased risk of getting the disease, with certain genes known to be inheritable risk factors. Environmental risks include exposure to pesticides and prior head injuries; a history of exposure to trichloroethylene is also suspected.
Diagnosis of Parkinson's disease is mainly based on symptoms, usually motor-related. PD typically occurs in people over the age of 60, of whom about one percent are affected. In those younger than 50, it is termed early-onset PD. The average post-diagnosis life expectancy is 7–15 years. No cure for PD is known, and treatment aims to mitigate symptoms. Initial treatment typically includes L-DOPA, MAO-B inhibitors, or dopamine agonists. As the disease progresses, these medications become less effective and produce a side effect marked by involuntary muscle movements. Diet and certain forms of rehabilitation have shown some effectiveness at improving symptoms. Surgery to place microelectrodes for deep brain stimulation has been used to reduce severe motor symptoms where drugs are ineffective. Evidence for treatments for the non-movement-related symptoms of PD, such as sleep disturbances and emotional problems, is less strong.
The disease is named after English doctor James Parkinson, who published the first detailed description in An Essay on the Shaking Palsy, in 1817. Public awareness campaigns include World Parkinson's Day and the use of a red tulip symbolizes Parkinson's awareness. People with PD who have increased the public's awareness of the condition include boxer Muhammad Ali and actor Michael J. Fox.
Classification edit
A progressive neurodegenerative disease,[9] Parkinson's disease (PD) is the most common form of parkinsonism—which encompasses tremor, bradykinesia, rigidity, and postural instability—and is also called idiopathic parkinsonism, meaning that it has no identifiable cause.[10]
Parkinson's can be classified in other ways. The parkinsonian syndromes include Parkinson's, along with other conditions.[11] It is included among the Lewy body diseases[12] and is a synucleinopathy, meaning that it is characterized by abnormal deposits of alpha-synuclein protein in the brain. The synucleinopathies include dementia with Lewy bodies, multiple system atrophy, and other rarer conditions.[13][9] Some rare genetic forms of Parkinson's do not exhibit alpha-synuclein aggregation.[citation needed]
Signs and symptoms edit
The most recognizable symptoms are movement (motor) related and include tremor, bradykinesia (slowness of movement), rigidity, and shuffling/stooped gait.[14] Non-motor symptoms, including autonomic dysfunction (dysautonomia), neuropsychiatric problems (mood, cognition, behavior or thought alterations), and sensory (especially altered sense of smell) and sleep difficulties may be present as well. People with Parkinson's disease may have non-motor symptoms that precede the onset of motor symptoms, including constipation, anosmia (inability to smell), and REM Behavior Disorder. Generally, symptoms such as dementia, psychosis, orthostasis, and more severe falls occur later.[14] The prevalence of oropharyngeal dysphagia in PD is estimated to be as high as 82%. Dysphagia (swallowing difficulties) has been reported across all stages of the disease. Complications result from dysphagia include dehydration, malnutrition, weight loss, and aspiration pneumonia. Pneumonia is the most common cause of hospitalization and a leading cause of death in people with PD.[15][16]
Motor edit
Four motor symptoms are considered as cardinal signs in PD: tremor, slowness of movement (bradykinesia), rigidity, and postural instability.[14]
The most common presenting sign is a coarse, slow tremor of the hand at rest, which disappears during voluntary movement of the affected arm and in the deeper stages of sleep.[14] It typically appears in only one hand, eventually affecting both hands as the disease progresses.[14] Frequency of PD tremor is between 4–6 hertz (cycles per second). A common characteristic of tremor is pill-rolling, the tendency of the index finger and thumb to touch and perform together with a circular movement.[14][17] The term derives from the similarity between the movement of people with PD and the early pharmaceutical technique of manually making pills.[17]
Bradykinesia is due to disturbances in motor planning of movement initiation, and associated with difficulties along the whole course of the movement process, from planning to initiation to execution of a movement. Performance of sequential and simultaneous movement is impaired. Bradykinesia is the most handicapping symptom of Parkinson's disease, presenting as difficulties with everyday tasks such as dressing, feeding, and bathing. It leads to particular difficulty in carrying out two independent motor activities at the same time, and can be made worse by emotional stress or concurrent illnesses. Paradoxically, people with PD can ride a bicycle or climb stairs more easily than walk on the level. Although most physicians may readily notice bradykinesia, formal assessment requires persons to do repetitive movements with their fingers and feet.[18]
In parkinsonism, rigidity or hypokinesia can be uniform, known as lead-pipe rigidity, or ratcheted, known as cogwheel rigidity.[10][14] The combination of tremor and increased tone is considered to be at the origin of cogwheel rigidity.[19] Rigidity may be associated with joint pain; such pain being a frequent initial manifestation of the disease.[14] In early stages of PD, rigidity is asymmetrical and tends to affect the neck and shoulder muscles before the muscles of the face and extremities.[20] With the progression of the disease, rigidity typically affects the whole body and reduces the ability to move.
Postural instability is typical in the later stages of the disease, leading to impaired balance and frequent falls,[21] and secondarily to bone fractures, loss of confidence, and reduced mobility.[22] Instability is absent in the initial stages, especially in younger people, especially before the development of bilateral symptoms.[23] Up to 40% of people diagnosed with PD may experience falls, and around 10% may have falls weekly, with the number of falls being related to the severity of PD.[14]
Other recognized motor signs and symptoms include gait and posture disturbances such as festination (rapid shuffling steps and a forward-flexed posture when walking with no flexed arm swing). Other common signs include freezing of gait (brief arrests when the feet seem to get stuck to the floor, especially on turning or changing direction), a slurred, monotonous, quiet voice, mask-like facial expression, and handwriting that gets smaller and smaller.[24] Although individuals with PD typically have motor symptoms predominantly on one side of the body, mask-like facial expression can occur on both sides of the face.[25]
Cognitive edit
PD causes neuropsychiatric disturbances ranging from mild to severe including disorders of cognition, mood, behavior, and thought.[14] Cognitive disturbances can occur in the early stages or before diagnosis, and increase in prevalence with duration of the disease.[14][26] The most common cognitive deficit is executive dysfunction, which can include problems with planning, cognitive flexibility, abstract thinking, rule acquisition, inhibiting inappropriate actions, initiating appropriate actions, working memory, and control of attention.[26][27] Other cognitive difficulties include slowed cognitive processing speed, impaired recall, and impaired perception and estimation of time.[26][27] Nevertheless, improvement appears when recall is aided by cues.[26] Visuospatial difficulties are a part of the disease, seen for example when the individual is asked to perform tests of facial recognition and perception of the orientation of drawn lines.[26][27]
Psychosis edit
Psychosis can be considered a symptom with a prevalence from 26 to 83%.[28][29] Hallucinations or delusions occur in about 50% of people with PD over the course of the illness, and may herald the emergence of dementia. These range from minor hallucinations – sense of passage (something quickly passing beside the person) or sense of presence (the perception of something or someone standing to the side or behind the person) – to full blown vivid, formed visual hallucinations and paranoid ideation. Auditory hallucinations are uncommon in PD, and are rarely described as voices. Psychosis is believed to be an integral part of the disease. A psychosis with delusions and associated delirium is a recognized complication of anti-Parkinson drug treatment. Urinary tract infections (frequent in the elderly) and underlying brain pathology or changes in neurotransmitters or their receptors (e.g., acetylcholine, serotonin) are thought to play a role in psychosis in PD.[30][31]
Neuropsychiatric edit
Behavior and mood alterations are more common in PD without cognitive impairment than in the general population and are usually present in PD with dementia. The most frequent mood difficulties are depression, apathy, and anxiety.[14] Depression impacts an estimated 20% to 35% of patients, and may appear at any stage of the disease. It can manifest with symptoms common to the disease process (fatigue, insomnia, and difficulty with concentration), which makes diagnosis difficult. The imbalance and changes in dopamine, serotonin, and noradrenergic hormones and functional impairment are causes of depression in PD-affected people.[28][32] Suicidal ideation is higher than in the general population, but suicidal attempts themselves are lower.[28][32] Risk factors for depression include disease onset under age 50, being female, previous history of depression, or severe motor symptoms.[28]
Anxiety has been estimated to have a prevalence in PD-affected people usually around 30–40% and up to 60% has been found.[28][32] Anxiety with PD is complex and consists of symptoms specific to PD.[33][page needed] Anxiety can be higher during motor "off" periods (times when medication is ineffective) and is likely diagnosed after diagnosis due to dysfunction of neurotransmitter pathways.[33][page needed] PD-affected people experience panic attacks more frequently compared with the general population. Both anxiety and depression have been found to be associated with decreased quality of life.[28][34] Symptoms can range from mild and episodic to chronic with potential causes being abnormal gamma-aminobutyric acid levels and embarrassment or fear about symptoms or disease.[28][34] Risk factors for anxiety in PD are disease onset under age 50, women, and off periods.[28]
Apathy and anhedonia are defined as a loss of motivation and an impaired ability to experience pleasure[35] and are symptoms classically associated with depression, but differ in PD-affected people in treatment and mechanism. Apathy presents in around 16.5–40%. Symptoms of apathy include reduced initiative/interests in new activities or the world around them, emotional indifference, and loss of affection or concern for others.[28] Apathy is associated with deficits in cognitive functions including executive and verbal memory.[32] Anhedonia occurs in 5–75% of people with PD, depending on the study population assessed and overlap with apathy.[36]
Impulse-control disorders, including pathological gambling, compulsive sexual behavior, binge eating, compulsive shopping, and reckless generosity, can be medication-related, particularly orally active dopamine agonists. The dopamine dysregulation syndrome – with wanting of medication contributing to overuse – is a rare complication of levodopa use.[37]
Gastrointestinal edit
Gastrointestinal issues in Parkinson's disease include constipation, impaired stomach emptying (gastric dysmotility), and excessive production of saliva can be severe enough to cause discomfort or endanger health.[38][39] Other upper gastrointestinal symptoms include swallowing impairment (Oropharyngeal dysphagia) and small intestinal bacterial overgrowth.[40]
Individuals with Parkinson's have alpha-synuclein deposits in the digestive tract as well as the brain.[40] Constipation is one of the symptoms associated with an increased risk of PD and may precede diagnosis of PD.[40]
Other edit
Sleep disorders occur with PD and can be worsened by medications.[14] Symptoms can manifest as daytime drowsiness (including sudden sleep attacks resembling narcolepsy), disturbances in rapid eye movement sleep, or insomnia.[14] REM behavior disorder may begin years before the development of motor or cognitive elements of PD or dementia with Lewy bodies.[41]
Alterations in the autonomic nervous system can lead to orthostatic hypotension (low blood pressure on standing), oily skin, excessive sweating, urinary incontinence, and altered sexual function.[14]
Changes in perception may include an impaired sense of smell, disturbed vision, pain, and paresthesia (tingling and numbness).[14] These symptoms can occur years before diagnosis of the disease.[14]
Causes and risk factors edit
The typical motor symptoms of Parkinson's are due to the deaths of dopaminergic neurons in the substantia nigra pars compacta (SNpc) brain region, resulting in a dopamine deficit in the striatum. Surviving neurons usually possess Lewy bodies—heavily composed of alpha-synuclein—which disrupt cellular processes.[42]
However, the underlying cause of Parkinson's is unknown,[43] and none of the proposed risk factors have been conclusively proven.[44] Age is the most significant risk factor.[45] The most frequently replicated environmental relationships are an increased risk in those exposed to pesticides and a reduced risk in smokers.[44][46] Research indicates that PD results from a complex interaction between genetic and environmental factors.[4] Only a minority of cases can be attributed solely to genetic causes. As idiopathic PD can last and progress over decades, the timing of exposure factor may influence the progression or severity of certain stages.[47]
Genetic edit
Research indicates that PD results from a complex interaction between genetic and environmental factors.[4][48] Around 15% of diagnosed individuals have a first-degree relative with the disease,[10] and 5–10% have a mutation in at least one high risk gene.[49][50] Harboring one of these gene mutations may not lead to the disease; susceptibility factors put them at an increased risk, in combination with other factors, which affect age of onset, severity and progression.[49]
As of 2022, around 90 genetic risk loci have been identified.[51][52] Many of these genes are known to contribute to cellular processes like cell death and inflammation, as well as the dysfunction of organelles like mitochondria, lysosomes, and endosomes.[51] The greatest genetic risk factor for Parkinson's is a heterozygous mutation in the enzyme-encoding GBA1 gene. It is found in 5–10% of those with PD.[53] A few genes are alone able to cause "monogenic" Parkinson's, which comprise 10 to 20 percent of cases.[54]
Alpha-synuclein, a protein encoded by SNCA gene mutations, is the main component of the Lewy bodies that accumulate in the brains of people with PD.[49] Alpha-synuclein activates ataxia telangiectasia mutated, a major DNA damage-repair signaling kinase.[55] In addition, alpha-synuclein activates the non-homologous end joining DNA repair pathway. The aggregation of alpha-synuclein in Lewy bodies appears to be a link between reduced DNA repair and brain-cell death in PD.[55] Major genetic risk factors that do not contribute to Lewy bodies include LRRK2 and PRKN.[56]
Non-genetic edit
Although the vast majority or Parkinson's cases are believed to be non-genetic, identifying environmental risk factors and causality is extremely difficult due to the disease's often decade-long prodromal period.[47] However, multi-decade studies have identified an increased likelihood of Parkinson's in association with agricultural work, pesticide exposure, and rural habitation.[57] In particular, exposure to pesticides like paraquat, rotenone, benomyl, and mancozeb are associated with the onset of Parkinson's disease.[58][59] The neurotoxic mechanisms for these pesticides have largely been identified: inhibition of the mitochondrial electron transport chain, increased oxidative stress, or inhibition of enzymes like aldehyde dehydrogenase and acetylcholinesterase.[60] For most patients, consumption of pesticide-treated food products is believed to be the primary source of exposure.[61]
Some medical drugs are implicated in parkinsonism; drug-induced parkinsonism is normally reversible by stopping the offending agent,[62] such as phenothiazines, butyrophenones, metoclopramide, and Tetrabenazine. MPTP is a drug known for causing irreversible parkinsonism: consequently, it is commonly used in Parkinson's animal-model research.[62][63][64] Low concentrations of urate in the blood are associated with an increased risk.[65] Moreover, a possible link exists between PD and Helicobacter pylori infection that can prevent the absorption of some drugs, including levodopa.[66][67]
Chlorinated solvents, used in commercial and industrial application like dry cleaning and degreasing, are associated with increased PD risk, particularly trichloroethylene.[68][57] Other chemical risk factors include manganese, suspended particles from traffic fumes, and exposure to other heavy metals such as mercury and lead.[57][69] Traumatic brain injuries (TBIs) are also strongly implicated as risk factors for PD.[70] Additionally, although the underlying cause is unknown, melanoma is a widely-documented risk factor for PD.[71]
Pathophysiology edit
The main pathological characteristics of PD are cell death in the brain's basal ganglia (affecting up to 70% of the dopamine-secreting neurons in the substantia nigra pars compacta by the end of life).[72] In Parkinson's disease, alpha-synuclein becomes misfolded and clump together with other alpha-synuclein. Cells are unable to remove these clumps, and the alpha-synuclein becomes cytotoxic, damaging the cells.[73][74] These clumps can be seen in neurons under a microscope and are called Lewy bodies. Loss of neurons is accompanied by the death of astrocytes (star-shaped glial cells) and an increase in the number of microglia (another type of glial cell) in the substantia nigra.[75][page needed] Severity of progression of the parts of the brain affected by PD can be measured with Braak staging. According to this staging, PD starts in the medulla and the olfactory bulb before moving to the substantia nigra pars compacta and the rest of the midbrain/basal forebrain. Movement symptom onset is associated when the disease begins to affect the substantia nigra pars compacta.[76]
Five major pathways in the brain connect other brain areas to the basal ganglia. These are known as the motor, oculomotor, associative, limbic, and orbitofrontal circuits. Names indicate the main projection area of each circuit.[77] All are affected in PD, and their disruption causes movement-, attention- and learning-related symptoms of the disease.[77] Scientifically, the motor circuit has been examined the most intensively.[77]
Since 1980, a particular conceptual model of the motor circuit and its alteration with PD has been of influence although some limitations have been pointed out which have led to modifications.[77] In this model, the basal ganglia normally exert a constant inhibitory influence on a wide range of motor systems, preventing them from becoming active at inappropriate times. When a decision is made to perform a particular action, inhibition is reduced for the required motor system, thereby releasing it for activation. Dopamine acts to facilitate this release of inhibition, so high levels of dopamine function tend to promote motor activity, while low levels of dopamine function, such as occur in PD, demand greater exertions of effort for any given movement. The result of dopamine depletion is to produce hypokinesia, an overall reduction in motor output.[77] Drugs that are used to treat PD, conversely, may produce excessive dopamine activity, allowing motor systems to be activated at inappropriate times and thereby producing dyskinesias.[77]
Brain cell death edit
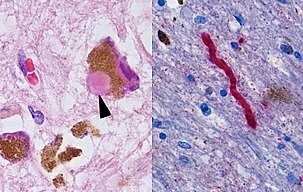
One mechanism causing brain cell death results from abnormal accumulation of the protein alpha-synuclein bound to ubiquitin in damaged cells. This insoluble protein accumulates inside neurons forming inclusions, known as Lewy bodies.[72][78] These bodies first appear in the olfactory bulb, medulla oblongata and pontine tegmentum; individuals at this stage may be asymptomatic or have early nonmotor symptoms (such as loss of sense of smell or some sleep or automatic dysfunction). As the disease progresses, Lewy bodies develop in the substantia nigra, areas of the midbrain and basal forebrain, and finally, the neocortex.[72] These brain sites are the main places of neuronal degeneration in PD, but Lewy bodies may be protective from cell death (with the abnormal protein sequestered or walled off). Other forms of alpha-synuclein (e.g. oligomers) that are not aggregated into Lewy bodies and Lewy neurites, may in fact be the toxic forms of the protein.[79][78] In people with dementia, a generalized presence of Lewy bodies is common in cortical areas. Neurofibrillary tangles and senile plaques, characteristic of Alzheimer's disease, are uncommon unless the person has dementia.[75][page needed]
Other mechanisms include proteasomal and lysosomal systems dysfunction and reduced mitochondrial activity.[79] Iron accumulation in the substantia nigra is typically observed in conjunction with the protein inclusions. It may be related to oxidative stress, protein aggregation, and neuronal death, but the mechanisms are obscure.[80]
Neuroimmune interaction edit
The neuroimmune interaction is heavily implicated in PD pathology. PD and autoimmune disorders share genetic variations and molecular pathways. Some autoimmune diseases may even increase one's risk of developing PD, up to 33% in one study.[81] Autoimmune diseases linked to protein expression profiles of monocytes and CD4+ T cells are linked to PD. Herpes virus infections can trigger autoimmune reactions to alpha-synuclein, perhaps through molecular mimicry of viral proteins.[82] Alpha-synuclein, and its aggregate form, Lewy bodies, can bind to microglia. Microglia can proliferate and be over-activated by alpha-synuclein binding to MHC receptors on inflammasomes, bringing about a release of proinflammatory cytokines like IL-1β, IFNγ, and TNFα.[83]
Activated microglia influence the activation of astrocytes, converting their neuroprotective phenotype to a neurotoxic one. Astrocytes in healthy brains serve to protect neuronal connections. In Parkinson's disease, astrocytes cannot protect the dopaminergic connections in the striatum. Microglia present antigens via MHC-I and MHC-II to T cells. CD4+ T cells, activated by this process, are able to cross the blood brain barrier (BBB) and release more proinflammatory cytokines, like interferon-γ (IFNγ), TNFα, and IL-1β. Mast cell degranulation and subsequent proinflammatory cytokine release is implicated in BBB breakdown in PD. Another immune cell implicated in PD are peripheral monocytes and have been found in the substantia nigra of people with PD. These monocytes can lead to more dopaminergic connection breakdown. In addition, monocytes isolated from people with Parkinson's disease express higher levels of the PD-associated protein, LRRK2, compared with non-PD individuals via vasodilation.[84] In addition, high levels of pro-inflammatory cytokines, such as IL-6, can lead to the production of C-reactive protein by the liver, another protein commonly found in people with PD, that can lead to an increase in peripheral inflammation.[85][86]
Peripheral inflammation can affect the gut-brain axis, an area of the body highly implicated in PD. People with PD have altered gut microbiota and colon problems years before motor issues arise.[85][86] Alpha-synuclein is produced in the gut and may migrate via the vagus nerve to the brainstem, and then to the substantia nigra.[undue weight? ][better source needed][87]
Diagnosis edit
Physician's initial assessment is typically based on medical history and neurological examination.[14] They assess motor symptoms (bradykinesia, rest tremors, etc.) using clinical diagnostic criteria. The finding of Lewy bodies in the midbrain on autopsy is usually considered final proof that the person had PD. The clinical course of the illness over time may diverge from PD, requiring that presentation is periodically reviewed to confirm the accuracy of the diagnosis.[14][88]
Multiple causes can occur for parkinsonism or diseases that look similar. Stroke, certain medications, and toxins can cause "secondary parkinsonism" and need to be assessed during visit.[76][88] Parkinson-plus syndromes, such as progressive supranuclear palsy and multiple system atrophy, must be considered and ruled out appropriately to begin a different treatment and disease progression (anti-Parkinson's medications are typically less effective at controlling symptoms in Parkinson-plus syndromes).[14] Faster progression rates, early cognitive dysfunction or postural instability, minimal tremor, or symmetry at onset may indicate a Parkinson-plus disease rather than PD itself.[89]
Medical organizations have created diagnostic criteria to ease and standardize the diagnostic process, especially in the early stages of the disease. The most widely known criteria come from the UK Queen Square Brain Bank for Neurological Disorders and the U.S. National Institute of Neurological Disorders and Stroke. The Queen Square Brain Bank criteria require slowness of movement (bradykinesia) plus either rigidity, resting tremor, or postural instability. Other possible causes of these symptoms need to be ruled out. Finally, three or more of the following supportive symptoms are required during onset or evolution: unilateral onset, tremor at rest, progression in time, asymmetry of motor symptoms, response to levodopa for at least five years, the clinical course of at least ten years and appearance of dyskinesias induced by the intake of excessive levodopa.[90] Assessment of sudomotor function through electrochemical skin conductance can be helpful in diagnosing dysautonomia.[91]
When PD diagnoses are checked by autopsy, movement disorders experts are found on average to be 79.6% accurate at initial assessment and 83.9% accurate after refining diagnoses at follow-up examinations. When clinical diagnoses performed mainly by nonexperts are checked by autopsy, the average accuracy is 73.8%. Overall, 80.6% of PD diagnoses are accurate, and 82.7% of diagnoses using the Brain Bank criteria are accurate.[92]
Imaging edit
Computed tomography (CT) scans of people with PD usually appear normal.[93] Magnetic resonance imaging has become more accurate in diagnosis of the disease over time, specifically through iron-sensitive T2* and susceptibility weighted imaging sequences at a magnetic field strength of at least 3T, both of which can demonstrate absence of the characteristic 'swallow tail' imaging pattern in the dorsolateral substantia nigra.[94] In a meta-analysis, absence of this pattern was highly sensitive and specific for the disease.[95] A meta-analysis found that neuromelanin-MRI can discriminate individuals with Parkinson's from healthy subjects.[96] Diffusion MRI has shown potential in distinguishing between PD and Parkinson-plus syndromes, as well as between PD motor subtypes,[97] though its diagnostic value is still under investigation.[93] CT and MRI are used to rule out other diseases that can be secondary causes of parkinsonism, most commonly encephalitis and chronic ischemic insults, as well as less-frequent entities such as basal ganglia tumors and hydrocephalus.[93]
The metabolic activity of dopamine transporters in the basal ganglia can be directly measured with positron emission tomography and single-photon emission computed tomography scans. It has shown high agreement with clinical diagnoses of PD.[98] Reduced dopamine-related activity in the basal ganglia can help exclude drug-induced Parkinsonism. This finding is nonspecific and can be seen with both PD and Parkinson-plus disorders.[93] In the United States, DaTSCANs are only FDA approved to distinguish PD or Parkinsonian syndromes from essential tremor.[99]
Iodine-123-meta-iodobenzylguanidine myocardial scintigraphy can help locate denervation of the muscles of the heart which can support a PD diagnosis.[76]
Differential diagnosis edit
Secondary parkinsonism – The multiple causes of parkinsonism can be differentiated through careful history, physical examination, and appropriate imaging.[76][100] Other Parkinson-plus syndromes can have similar movement symptoms but have a variety of associated symptoms. Some of these are also synucleinopathies. Lewy body dementia involves motor symptoms with early onset of cognitive dysfunction and hallucinations which precede motor symptoms. Alternatively, multiple systems atrophy or MSA usually has early onset of autonomic dysfunction (such as orthostasis), and may have autonomic predominance, cerebellar symptom predominance, or Parkinsonian predominance.[101]
Other Parkinson-plus syndromes involve tau, rather than alpha-synuclein. These include progressive supranuclear palsy (PSP) and corticobasal syndrome (CBS). PSP predominantly involves rigidity, early falls, bulbar symptoms, and vertical gaze restriction; it can be associated with frontotemporal dementia symptoms. CBS involves asymmetric parkinsonism, dystonia, alien limb, and myoclonic jerking.[102] Presentation timelines and associated symptoms can help differentiate similar movement disorders from idiopathic Parkinson disease.[medical citation needed]
Vascular parkinsonism is the phenomenon of the presence of Parkinson's disease symptoms combined with findings of vascular events (such as a cerebral stroke). The damaging of the dopaminergic pathways is similar in cause for both vascular parkinsonism and idiopathic PD, and so present with similar symptoms. Differentiation can be made with careful bedside examination, history evaluation, and imaging.[103][104]
Parkinson-plus syndrome – Multiple diseases can be considered part of the Parkinson's plus group, including corticobasal syndrome, multiple system atrophy, progressive supranuclear palsy, and dementia with Lewy bodies. Differential diagnosis can be narrowed down with careful history and physical exam (especially focused on the sequential onset of specific symptoms), progression of the disease, and response to treatment.[105][100] Some key symptoms:[62][100]
- Corticobasal syndrome – levodopa-resistance, myoclonus, dystonia, corticosensory loss, apraxia, and non-fluent aphasia
- Dementia with Lewy bodies – levodopa resistance, cognitive predominance before motor symptoms, and fluctuating cognitive symptoms, (visual hallucinations are common in this disease)
- Essential tremor – This can at first look like parkinsonism, but has key differentiators. In essential tremor, the tremor gets worse with action (improves in PD), a lack of other symptoms is common in PD, and normal DatSCAN is seen.[100]
- Multiple system atrophy – levodopa resistance, rapidly progressive, autonomic failure, stridor, present Babinski sign, cerebellar ataxia, and specific MRI findings
- Progressive supranuclear palsy – levodopa resistance, restrictive vertical gaze, specific MRI findings, and early and different postural difficulties
Other conditions that can have similar presentations to PD include:[100][62]
- Arthritis
- Creutzfeldt–Jakob disease
- Depression
- Dystonia
- Fragile X-associated tremor/ataxia syndrome
- Frontotemporal dementia and parkinsonism linked to chromosome 17
- Huntington's disease
- Idiopathic basal ganglia calcification
- Neurodegeneration with brain iron accumulation
- Normal-pressure hydrocephalus
- Obsessional slowness
- Psychogenic parkinsonism
- Wilson's disease
Prevention edit
Exercise in middle age may reduce the risk of PD later in life.[106] A review of studies regarding how physical activity (exercise) prevents Parkinson's disease noted a 34% reduction in risk amongst individuals who perform moderate to vigorous physical activity. The potential benefits of exercise in individuals with Parkinson's disease is being researched.[107]
Caffeine appears protective with a greater decrease in risk occurring with a larger intake of caffeinated beverages such as coffee.[108]
Antioxidants, such as vitamins C and E, have been proposed to protect against the disease, but results of studies have been contradictory and no positive effect has been shown.[44] The results regarding fat and fatty acids have been contradictory.[44] Use of nonsteroidal anti-inflammatory drugs (NSAIDs) and calcium channel blockers may be protective.[4] A 2010 meta-analysis found that NSAIDs (apart from aspirin), have been associated with at least a 15% (higher in long-term and regular users) reduction in the incidence of the development of PD.[109] As of 2019[update] meta-analyses have failed to confirm this link. Multiple studies have demonstrated a link between the use of ibuprofen and a decreased risk of Parkinson's development.[110]
Management edit
Despite ongoing research efforts, Parkinson's disease has no known cure. Patients are typically managed by a holistic approach that combines lifestyle modifications with physical therapy. Certain medications may also be used to provide symptomatic relief and improve the patient's quality of life. The medications used in Parkinson's disease work by either increasing endogenous dopamine levels or by directly mimicking dopamine's effect on the patient's brain. Levodopa (L-dopa) is the most effective for motor symptoms and is often combined with a dopa decarboxylase inhibitor (such as carbidopa or benserazide).[111] Other agents include COMT inhibitors, dopamine agonists, and MAO-B inhibitors. The patient's age at disease onset as well as the stage of the disease often help determine which of the aforementioned drug groups is most effective.
Braak staging classifies the degree of pathology in patients with Parkinson's disease into six stages that correlate with early, middle, and late presentations of the disease.[112] Treatment in the first stage aims for an optimal trade-off between symptom control and medication side effects. For example, the administration of levodopa in this stage may be delayed in favor of other agents, such as MAO-B inhibitors and dopamine agonists, due to its notable risk of complications.[113] However, levodopa-related dyskinesias still correlate more strongly with the duration and severity of the disease than the duration of levodopa treatment.[114] A careful consideration of the risks and benefits of starting treatment is crucial to optimize patient outcomes. In the middle stages of Parkinson's disease, the primary aim is to reduce patient symptoms. Episodes of medication overuse or any sudden withdrawals from medication must be promptly managed.[113] If medications prove ineffective, surgery (such as deep brain stimulation or high-intensity focused ultrasound[115]), subcutaneous waking-day apomorphine infusion, and enteral dopa pumps may be useful.[116] Late-stage Parkinson's disease presents challenges requiring a variety of treatments, including those for psychiatric symptoms particularly depression, orthostatic hypotension, bladder dysfunction, and erectile dysfunction.[116] In the final stages of the disease, palliative care is provided to improve a person's quality of life.[117]
A 2020 Cochrane review found no certain evidence that cognitive training is beneficial for people with Parkinson's disease, dementia or mild cognitive impairment.[118] The findings are based on low certainty evidence of seven studies.
No standard treatment for PD-associated anxiety exists.[33][page needed]
Medications edit
Levodopa edit
Levodopa is usually the first drug of choice when treating Parkinson's disease and has been the most widely used PD treatment since the 1980s.[113][119] The motor symptoms of PD are the result of reduced dopamine production in the brain's basal ganglia. Dopamine fails to cross the blood–brain barrier so it cannot be taken as a medicine to boost the brain's depleted levels of dopamine. A precursor of dopamine, levodopa, can pass through to the brain where it is readily converted to dopamine. Administration of levodopa temporarily diminishes the motor symptoms of PD.
Only 5–10% of levodopa crosses the blood–brain barrier. Much of the remainder is metabolized to dopamine elsewhere in the body, causing a variety of side-effects, including nausea, vomiting, and orthostatic hypotension.[120] Carbidopa and benserazide are dopa decarboxylase inhibitors that fail to cross the blood–brain barrier and inhibit the conversion of levodopa to dopamine outside the brain, reducing side-effects and improving the availability of levodopa for passage into the brain. One of these drugs is usually taken along with levodopa and is available combined with levodopa in the same pill.[121]
Prolonged use of levodopa is associated with the development of complications, such as involuntary movements (dyskinesias) and fluctuations in the impact of the medication.[113] When fluctuations occur, a person can cycle through phases with good response to medication and reduced PD symptoms ("on state"), and phases with poor response to medication and increased PD symptoms ("off state").[113][122] Using lower doses of levodopa may reduce the risk and severity of these levodopa-induced complications.[123] A former strategy, called "drug holidays", to reduce levodopa-related dyskinesia and fluctuations was to withdraw levodopa medication for some time,[119] which can bring on dangerous side-effects such as neuroleptic malignant syndrome and is discouraged.[113] Most people with PD eventually need levodopa and later develop levodopa-induced fluctuations and dyskinesias.[113] Adverse effects of levodopa, including dyskinesias, may mistakenly influence people affected by the disease and sometimes their healthcare professionals to delay treatment, which reduces potential for optimal results.[medical citation needed]
Levodopa is available in oral, inhalation, and infusion form; inhaled levodopa can be used when oral levodopa therapy has reached a point where "off" periods have increased in length.[124][125]
COMT inhibitors edit
During the course of PD, affected people can experience a wearing-off phenomenon, where a recurrence of symptoms occurs after a dose of levodopa, but right before their next dose.[76] Catechol-O-methyltransferase (COMT) is a protein that degrades levodopa before it can cross the blood–brain barrier and COMT inhibitors allow for more levodopa to cross.[127] They are normally used in the management of later symptoms, but can be used in conjunction with levodopa/carbidopa when a person is experiencing the wearing off-phenomenon with their motor symptoms.[76][119]
Three COMT inhibitors are used to treat adults with PD and end-of-dose motor fluctuations – opicapone, entacapone, and tolcapone.[76] Tolcapone has been available for but its usefulness is limited by possible liver damage complications requiring liver-function monitoring.[128][76][127][62] Entacapone and opicapone cause little alteration to liver function.[127][129][130] Licensed preparations of entacapone contain entacapone alone or in combination with carbidopa and levodopa.[131][62][132] Opicapone is a once-daily COMT inhibitor.[133][76]
Dopamine agonists edit
Dopamine agonists that bind to dopamine receptors in the brain have similar effects to levodopa.[113] These were initially used as a complementary therapy to levodopa for individuals experiencing levodopa complications (on-off fluctuations and dyskinesias); they are mainly used on their own as first therapy for the motor symptoms of PD with the aim of delaying the initiation of levodopa therapy, thus delaying the onset of levodopa's complications.[113][134] Dopamine agonists include bromocriptine, pergolide, pramipexole, ropinirole, piribedil, cabergoline, apomorphine, and lisuride.
Though dopamine agonists are less effective than levodopa at controlling PD motor symptoms, they are effective enough to manage these symptoms in the first years of treatment.[10] Dyskinesias due to dopamine agonists are rare in younger people who have PD, but along with other complications, become more common with older age at onset.[10] Thus, dopamine agonists are the preferred initial treatment for younger-onset PD, and levodopa is preferred for older-onset PD.[10]
Dopamine agonists produce side effects, including drowsiness, hallucinations, insomnia, nausea, and constipation.[113][119] Side effects appear with minimal clinically effective doses giving the physician reason to search for a different drug.[113] Agonists have been related to impulse-control disorders (such as increased sexual activity, eating, gambling, and shopping) more strongly than other antiparkinson medications.[135][119]
Apomorphine, a dopamine agonist, may be used to reduce off periods and dyskinesia in late PD.[113] It is administered only by intermittent injections or continuous subcutaneous infusions.[113] Secondary effects such as confusion and hallucinations are common, individuals receiving apomorphine treatment should be closely monitored.[113] Two dopamine agonists administered through skin patches (lisuride and rotigotine) are useful for people in the initial stages and possibly to control off states in those in advanced states.[136][page needed] Due to an increased risk of cardiac fibrosis with ergot-derived dopamine agonists (bromocriptine, cabergoline, dihydroergocryptine, lisuride, and pergolide), they should only be considered for adjunct therapy to levodopa.[119]
MAO-B inhibitors edit
MAO-B inhibitors (safinamide, selegiline and rasagiline) increase the amount of dopamine in the basal ganglia by inhibiting the activity of monoamine oxidase B, an enzyme that breaks down dopamine.[113] They have been found to help alleviate motor symptoms when used as monotherapy (on their own); when used in conjunction with levodopa, time spent in the off phase is reduced.[137][119] Selegiline has been shown to delay the need for beginning levodopa, suggesting that it might be neuroprotective and slow the progression of the disease.[138] An initial study indicated that selegiline in combination with levodopa increased the risk of death, but this has been refuted.[139]
Common side effects are nausea, dizziness, insomnia, sleepiness, and (in selegiline and rasagiline) orthostatic hypotension.[138][76] MAO-Bs are known to increase serotonin and cause a potentially dangerous condition known as serotonin syndrome.[138]
Other drugs edit
Other drugs such as amantadine may be useful as treatment of motor symptoms, but evidence for use is lacking.[113][140] Anticholinergics should not be used for dyskinesia or motor fluctuations but may be considered topically for drooling.[119] A diverse range of symptoms beyond those related to motor function can be treated pharmaceutically.[141] Examples are the use of quetiapine or clozapine for psychosis, cholinesterase inhibitors or memantine for dementia, and modafinil for excessive daytime sleepiness.[141][142][119] In 2016, pimavanserin was approved for the management of PD psychosis.[143] Doxepin and rasagline may reduce physical fatigue in PD.[144]
Surgery edit
Treating motor symptoms with surgery was once a common practice but the discovery of levodopa has decreased the amount of procedures.[145] Studies have led to great improvements in surgical techniques, so surgery can be used in people with advanced PD for whom drug therapy is no longer sufficient.[145] Surgery for PD can be divided in two main groups – lesional and deep brain stimulation (DBS). Target areas for DBS or lesions include the thalamus, globus pallidus, or subthalamic nucleus.[145] DBS involves the implantation of a medical device called a neurostimulator, which sends electrical impulses to specific parts of the brain. DBS is recommended for people who have PD with motor fluctuations and tremor inadequately controlled by medication, or to those who are intolerant to medication lacking severe neuropsychiatric problems.[146] Other less common surgical therapies involve intentional formation of lesions to suppress overactivity of specific subcortical areas. For example, pallidotomy involves surgical destruction of the globus pallidus to control dyskinesia.[145]
Four areas of the brain have been treated with neural stimulators in PD.[147] These are the globus pallidus interna, thalamus, subthalamic nucleus, and pedunculopontine nucleus. DBS of the globus pallidus interna improves motor function, while DBS of the thalamic DBS improves tremor, but has little impact on bradykinesia or rigidity. DBS of the subthalamic nucleus is usually avoided if a history of depression or neurocognitive impairment is present. DBS of the subthalamic nucleus is associated with a reduction in medication. Pedunculopontine nucleus DBS remains experimental at present. Generally, DBS is associated with 30–60% improvement in motor score evaluations.[147]
Rehabilitation edit
Exercise programs are recommended in people with PD and recent reviews (2023) have demonstrated their efficacy.[148][106] Some evidence shows that speech or mobility problems can improve with rehabilitation, although studies are scarce and of low quality.[149][150] Regular physical exercise with or without physical therapy can be beneficial to maintain and improve mobility, flexibility, strength, gait speed, and quality of life.[150] When an exercise program is performed under the supervision of a physiotherapist, more improvements occur in motor symptoms, mental and emotional functions, daily living activities, and quality of life compared with a self-supervised exercise program at home.[151] Clinical exercises may be an effective intervention targeting overall well-being of individuals with Parkinson's. Improvement in motor function and depression may happen.[152]
In improving flexibility and range of motion for people experiencing rigidity, generalized relaxation techniques such as gentle rocking have been found to decrease excessive muscle tension. Other effective techniques to promote relaxation include slow rotational movements of the extremities and trunk, rhythmic initiation, diaphragmatic breathing, and meditation techniques.[153] As for gait and addressing the challenges associated with the disease such as hypokinesia, shuffling, and decreased arm swing, physiotherapists have a variety of strategies to improve functional mobility and safety. Areas of interest concerning gait during rehabilitation programs focus on improving gait speed, the base of support, stride length, and trunk and arm-swing movement. Strategies include using assistive equipment (pole walking and treadmill walking), verbal cueing (manual, visual, and auditory), exercises (marching and PNF patterns), and altering environments (surfaces, inputs, open vs. closed).[154] Strengthening exercises have shown improvements in strength and motor function for people with primary muscular weakness and weakness related to inactivity with mild to moderate PD, but reports show an interaction between strength and the time the medications were taken. Therefore, people with PD should perform exercises 45 minutes to one hour after medications when they are capable.[155] Deep diaphragmatic breathing exercises are beneficial in improving chest-wall mobility and vital capacity decreased by a forward flexed posture and respiratory dysfunctions in advanced PD.[156] Exercise may improve constipation.[38] Whether exercise reduces physical fatigue in PD remains unclear.[144]
Strength training exercise has been shown to increase manual dexterity in people with PD after exercising with manual putty. Improvements in both manual dexterity and strength can have a favorable impact on people with Parkinson's disease, fostering increased independence in daily activities that require grasping objects.[157]
The Lee Silverman voice treatment (LSVT) is one of the most widely practiced treatments for speech disorders associated with PD.[149][158] Speech therapy and specifically LSVT may improve speech.[149] Occupational therapy (OT) aims to promote health and quality of life by helping people with the disease to participate in a large percentage of their daily living activities.[149] Few studies have been conducted on the effectiveness of OT, and their quality is poor, although with some indication that it may improve motor skills and quality of life for the duration of the therapy.[149][159]
Palliative care edit
The goal of Palliative care is to improve quality of life for both the patient and family by providing relief from the symptoms and stress of illnesses.[160] As Parkinson's is uncurable, treatments focus on slowing decline and improving quality of life and are therefore palliative.[161]
Palliative care should be involved earlier, rather than later, in the disease course.[162][163] Palliative care specialists can help with physical symptoms, emotional factors such as loss of function and jobs, depression, fear, and existential concerns.[162][163][164]
Along with offering emotional support to both the affected person and family, palliative care addresses goals of care. People with PD may have difficult decisions to make as the disease progresses, such as wishes for feeding tube, noninvasive ventilator or tracheostomy, wishes for or against cardiopulmonary resuscitation, and when to use hospice care.[161] Palliative-care team members can help answer questions and guide people with PD on these complex and emotional topics to help them make decisions based on values.[163][165]
Muscles and nerves that control the digestive process may be affected by PD, resulting in constipation and gastroparesis (prolonged emptying of stomach contents).[38] A balanced diet, based on periodical nutritional assessments, is recommended, and should be designed to avoid weight loss or gain and minimize the consequences of gastrointestinal dysfunction.[38] As the disease advances, swallowing difficulties (dysphagia) may appear. Using thickening agents for liquid intake and an upright posture when eating may be useful; both measures reduce the risk of choking. Gastrostomy can be used to deliver food directly into the stomach.[38]
Levodopa and proteins use the same transportation system in the intestine and the blood–brain barrier, thereby competing for access.[38] Taking them together results in reduced effectiveness of the drug.[38] Therefore, when levodopa is introduced, excessive protein consumption is discouraged in favour of a well-balanced Mediterranean diet. In advanced stages, additional intake of low-protein products such as bread or pasta is recommended for similar reasons.[38] To minimize interaction with proteins, levodopa should be taken 30 minutes before meals.[38] At the same time, regimens for PD restrict proteins during breakfast and lunch, allowing protein intake in the evening.[38]
Prognosis edit
PD invariably progresses with time. A severity rating method known as the Unified Parkinson's disease rating scale (UPDRS) is the most commonly used metric for a clinical study. A modified version known as the MDS-UPDRS is also used. An older scaling method known as the Hoehn and Yahr scale (originally published in 1967), and a similar scale known as the Modified Hoehn and Yahr scale, have been used. The Hoehn and Yahr scale defines five basic stages of progression.
Motor symptoms may advance aggressively in the early stages of the disease and more slowly later. Untreated, individuals are expected to lose independent ambulation after an average of eight years and be bedridden after 10 years. Medication has improved the prognosis of motor symptoms. In people taking levodopa, the progression time of symptoms to a stage of high dependency from caregivers may be over 15 years. Predicting what course the disease will take for a given individual is difficult.[166] Age is an appropriate predictor of disease progression. The rate of motor decline is greater in those with less impairment at the time of diagnosis, while cognitive impairment is more frequent in those who are over 70 years of age at symptom onset.[79]
No hard correlation between PD progression and PD disability as a whole has been found as of 2023; however, initial PD disability often mainly concerns the motor symptoms, whereas in later stages it is often mainly related to the non-motor symptoms of the disease. Therapies exist that can improve or alleviate these latter symptoms to some extent.[79][166] As the disease advances, disability is more related to motor symptoms that are uncontrollable by medication (such as difficulties with swallowing and dysartria, and gait and balance problems) and to levodopa-induced complications, which appear in up to 50% of individuals after five years of levodopa usage. Finally, after ten years most people with the disease have autonomic disturbances, sleep problems, mood alterations and cognitive decline; these symptoms, especially cognitive decline, greatly increase disability.[79][166]
The life expectancy of people with PD is reduced. Mortality ratios are around twice those of unaffected people. Cognitive decline and dementia, old age at onset, a more advanced disease state, and presence of swallowing problems are all mortality risk factors. A disease pattern mainly characterized by tremor as opposed to rigidity, though, predicts an improved survival. Death from aspiration pneumonia is twice as common in individuals with PD as in the healthy population.[166]
In 2016, PD resulted in about 211,000 deaths globally, an increase of 161% since 1990.[167] The overall death rate increased by 19% to 1.81 per 100,000 people during that time.[167]
A person with PD has two to six times the risk of dementia compared with the general population.[14][26] Up to 78% of people with PD have Parkinson's disease dementia.[168] Dementia is associated with a reduced quality of life in people with PD and their caregivers, increased mortality, and a higher probability of needing nursing home care.[26]
Epidemiology edit
PD is the second most common neurodegenerative disorder and affects approximately six million people globally.[43] As of 2021, Parkinson's was the fastest growing neurodegenerative disease globally in both death and prevalence.[54] The proportion in a population at a given time is about 0.3% in industrialized countries. PD is more common in the elderly and rates rise from 1% in those over 60 years of age to 4% of the population over 80.[44][169] The mean age of onset is around 60 years, although 5–10% begin between the ages of 20 and 50 is classified as young onset PD.[10] Males are affected at a ratio of around 3:2 compared with females.[4] PD may be less prevalent in those of African and Asian ancestry (including South Asians), although this finding is disputed.[44] The number of new diagnoses per year of PD is between 8–18 per 100,000 person–years.[44]
China is predicted to have nearly half of the Parkinson's disease population in the world in 2030.[170] By 2040 the number of patients is expected to grow to approximately 14 million people; this growth has been referred to as the Parkinson's pandemic.[171]
The prevalence of dementia increases with age, and to a lesser degree, duration of the disease.[172]
History edit
Early sources, including an Egyptian papyrus, an ayurvedic medical treatise, the Bible, and Galen's writings, describe symptoms resembling those of PD.[175] After Galen, no references unambiguously related to PD appear until the 17th century.[175] In the 17th and 18th centuries, Franciscus Sylvius, Hieronymus David Gaubius, John Hunter and Auguste François Chomel wrote about elements of the disease.[175][176][177]
Despite that the first systematic description of the disease is attributed to James Parkinson, the disease itself with all four cardinal signs (tremor, bradykinesia, rigor and postural instability) was described in 1690 by Ferenc Pápai Páriz, in a Hungarian medical text over 120 years before the classical description of James Parkinson. However, as it was published in Hungarian language, his book was ignored in the western medical literature. [178] [179] [180] [181]
In 1817, James Parkinson published his essay reporting six people with paralysis agitans.[182] An Essay on the Shaking Palsy described the characteristic resting tremor, abnormal posture and gait, paralysis and diminished muscle strength, and the way that the disease progresses over time.[183][184] Early neurologists who made further additions to the knowledge of the disease include Trousseau, Gowers, Kinnier Wilson and Erb, and Jean-Martin Charcot, whose studies between 1868 and 1881 increased the understanding of the disease.[182] Among other advances, Charcot made the distinction between rigidity, weakness and bradykinesia.[182] He championed the renaming of the disease in honor of James Parkinson.[182]
In 1912, Frederic Lewy described microscopic particles in affected brains, later named Lewy bodies.[182] In 1919, Konstantin Tretiakoff reported that the substantia nigra was the main cerebral structure affected, but this finding was rejected until it was confirmed by further studies published by Rolf Hassler in 1938.[182] The underlying biochemical changes in the brain were identified in the 1950s, due largely to the work of Arvid Carlsson on the neurotransmitter dopamine and Oleh Hornykiewicz on its role on PD.[185] In 1997, alpha-synuclein was found to be the main component of Lewy bodies by Spillantini, Trojanowski, Goedert, and others.[78]
Anticholinergics and surgery (lesioning of the corticospinal pathway or some of the basal ganglia structures) were the only treatments until the arrival of levodopa, which reduced their use dramatically.[176][186] Levodopa was first synthesized in 1911 by Casimir Funk, but it received little attention until the mid 20th century.[185] It entered clinical practice in 1967 and brought about a revolution in the management of PD.[185][187] By the late 1980s deep brain stimulation introduced by Alim Louis Benabid and colleagues at Grenoble, France, emerged as a possible treatment.[188]
An illustration of advanced Parkinson's disease by William Richard Gowers was published in 1886 in A Manual of Diseases of the Nervous System, based on 1879 photographs attributed to Albert Londe.[173][174] A new image was created in 2020 to represent diversity in levels of severity.[189][190]
Society and culture edit
Social impact edit
For some people with PD, masked facial expressions and difficulty moderating facial expressions of emotion or recognizing other people's facial expressions can impact social well-being.[25] As the condition progresses, tremor, other motor symptoms, difficulty communicating, or issues with mobility may interfere with social engagement, causing individuals with PD to feel isolated.[191] Public perception and awareness of PD symptoms such as shaking, hallucinating, slurring speech, and being off balance is lacking in some countries and can lead to stigma.[191]
Cost edit
The costs of PD to society are high; in 2007, the largest share of direct cost came from inpatient care and nursing homes, while the share coming from medication was substantially lower.[192] Indirect costs are high, due to reduced productivity and the burden on caregivers.[192] In addition to economic costs, PD reduces quality of life of those with the disease and their caregivers.[192]
A study based on 2017 data estimated the US economic PD burden at $51.9 billion, including direct medical costs of $25.4 billion and $26.5 billion in indirect and non-medical costs. The projected total economic burden surpasses $79 billion by 2037. These findings highlight the need for interventions to reduce PD incidence, delay disease progression, and alleviate symptom burden that may reduce the future economic burden of PD.[193]
Advocacy edit
The birthday of James Parkinson, 11 April, has been designated as World Parkinson's Day.[182] A red tulip was chosen by international organizations as the symbol of the disease in 2005; it represents the 'James Parkinson' tulip cultivar, registered in 1981 by a Dutch horticulturalist.[194]
Advocacy organizations include the National Parkinson Foundation, which has provided more than $180 million in care, research, and support services since 1982,[195] Parkinson's Disease Foundation, which has distributed more than $115 million for research and nearly $50 million for education and advocacy programs since its founding in 1957 by William Black;[196][197] the American Parkinson Disease Association, founded in 1961;[198] and the European Parkinson's Disease Association, founded in 1992.[199]
Notable cases edit
In the 21st century, the diagnosis of Parkinson's among notable figures has increased the public's understanding of the disorder.[200]
Actor Michael J. Fox was diagnosed with PD at 29 years old,[201] and has used his diagnosis to increase awareness of the disease.[202] To illustrate the effects of the disease, Fox has appeared without medication in television roles and before the United States Congress.[203] The Michael J. Fox Foundation, which he founded in 2000, has raised over $2 billion for Parkinson's research.[204] Boxer Muhammad Ali showed signs of PD when he was 38, but was undiagnosed until he was 42, and has been called the "world's most famous Parkinson's patient".[205] Whether he had PD or parkinsonism related to boxing is unresolved.[206][207] Cyclist and Olympic medalist Davis Phinney, diagnosed with Parkinson's at 40, started the Davis Phinney Foundation in 2004 to support PD research.[208][209] Deng Xiaoping, former paramount leader of China, had advanced Parkinson's disease.[210][211][212]
At the time of his suicide in 2014, Robin Williams, the American actor and comedian, had been diagnosed with PD,[213] but his autopsy revealed dementia with Lewy bodies, highlighting difficulties in accurate diagnosis.[213][214][215][216][217]
Clinical research edit
As of 2022[update], no disease-modifying drugs (drugs that target the causes or damage) are approved for Parkinson's, so this is a major focus of Parkinson's research.[218][219] Active research directions include the search for new animal models of the disease and studies of the potential usefulness of gene therapy, stem cell transplants, and neuroprotective agents.[220] To aid in earlier diagnosis, research criteria for identifying prodromal biomarkers of the disease have been established.[221]
Gene therapy edit
Gene therapy typically involves the use of a noninfectious virus[222] to shuttle genetic material into a part of the brain. Approaches have involved the expression of growth factors to prevent damage (Neurturin – a GDNF-family growth factor), and enzymes such as glutamic acid decarboxylase (GAD – the enzyme that produces GABA), tyrosine hydroxylase (the enzyme that produces L-DOPA) and catechol-O-methyl transferase (COMT – the enzyme that converts L-DOPA to dopamine). No safety concerns have been reported but the approaches have largely failed in phase two clinical trials.[220] The delivery of GAD showed promise in phase two trials in 2011, but while effective at improving motor function, was inferior to DBS. Follow-up studies in the same cohort have suggested persistent improvement.[223]
Neuroprotective treatments edit
A vaccine that primes the human immune system to destroy alpha-synuclein, PD01A, entered clinical trials and a phase one report in 2020 suggested safety and tolerability.[224][225] In 2018, an antibody, PRX002/RG7935, showed preliminary safety evidence in stage I trials supporting continuation to stage II trials.[226]
Cell-based therapies edit
In contrast to other neurodegenerative disorders, many Parkinson's symptoms can be attributed to the loss of a single cell type: mesencephalic dopaminergic (DA) neurons. Consequently, DA neuron regeneration is a promising therapeutic approach.[227] Although most initial research sought to generate DA neuron precursor cells from fetal brain tissue,[228] pluripotent stem cells—particularly induced pluripotent stem cells (iPSCs)—have become an increasingly popular tissue source.[229][230]
Both fetal and iPSC-derived DA neurons have been transplanted into patients in clinical trials.[231][232] Although some patients see improvements, the results are highly variable. Adverse effects, such as dyskinesia arising from excess dopamine release by the transplanted tissues, have also been observed.[233][234]
Pharmaceutical edit
Antagonists of adenosine receptors (specifically A2A) have been explored for Parkinson's.[235] Of these, istradefylline has emerged as the most successful medication and was approved for medical use in the United States in 2019.[236] It is approved as an add-on treatment to the levodopa/carbidopa regime.[236] LRRK2 kinase inhibitors are also of interest.[237]
References edit
Citations edit
- ^ a b c d "Parkinson's Disease Information Page". NINDS. 30 June 2016. Retrieved 18 July 2016.
- ^ a b Sveinbjornsdottir S (October 2016). "The clinical symptoms of Parkinson's disease". Journal of Neurochemistry. 139 (Suppl 1): 318–324. doi:10.1111/jnc.13691. PMID 27401947.
- ^ Truong & Bhidayasiri 2016, p. 188.
- ^ a b c d e f Kalia LV, Lang AE (August 2015). "Parkinson's disease". Lancet. 386 (9996): 896–912. doi:10.1016/s0140-6736(14)61393-3. PMID 25904081. S2CID 5502904.
- ^ Ferri 2010, Chapter P.
- ^ Vos T, Allen C, Arora M, Barber RM, Bhutta ZA, Brown A, et al. (GBD 2015 Disease and Injury Incidence and Prevalence Collaborators) (October 2016). "Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990–2015: a systematic analysis for the Global Burden of Disease Study 2015". Lancet. 388 (10053): 1545–1602. doi:10.1016/S0140-6736(16)31678-6. PMC 5055577. PMID 27733282.
- ^ Wang H, Naghavi M, Allen C, Barber RM, Bhutta ZA, Carter A, et al. (GBD 2015 Mortality and Causes of Death Collaborators) (October 2016). "Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980–2015: a systematic analysis for the Global Burden of Disease Study 2015". Lancet. 388 (10053): 1459–1544. doi:10.1016/s0140-6736(16)31012-1. PMC 5388903. PMID 27733281.
- ^ Connie T, Aderinola TB, Ong TS, Goh MKO, Erfianto B, Purnama B (12 December 2022). "Pose-Based Gait Analysis for Diagnosis of Parkinson's Disease". Algorithms. 15 (12). MDPI AG: 474. doi:10.3390/a15120474. ISSN 1999-4893.
- ^ a b Tulisiak CT, Mercado G, Peelaerts W, Brundin L, Brundin P (2019). "Can infections trigger alpha-synucleinopathies?". Prog Mol Biol Transl Sci. Progress in Molecular Biology and Translational Science. 168: 299–322. doi:10.1016/bs.pmbts.2019.06.002. ISBN 978-0-12-817874-4. PMC 6857718. PMID 31699323.
- ^ a b c d e f g Samii A, Nutt JG, Ransom BR (May 2004). "Parkinson's disease". Lancet. 363 (9423): 1783–1793. doi:10.1016/S0140-6736(04)16305-8. PMID 15172778. S2CID 35364322.
- ^ Levin J, Kurz A, Arzberger T, Giese A, Höglinger GU (February 2016). "The differential diagnosis and treatment of atypical parkinsonism". Dtsch Ärztebl Int (Review). 113 (5): 61–69. doi:10.3238/arztebl.2016.0061. PMC 4782269. PMID 26900156.
- ^ Kon T, Tomiyama M, Wakabayashi K (February 2020). "Neuropathology of Lewy body disease: Clinicopathological crosstalk between typical and atypical cases". Neuropathology. 40 (1): 30–39. doi:10.1111/neup.12597. PMID 31498507. S2CID 201983865.
- ^ Goedert M, Jakes R, Spillantini MG (2017). "The synucleinopathies: Twenty years on". J Parkinsons Dis (Review). 7 (s1): S51–S71. doi:10.3233/JPD-179005. PMC 5345650. PMID 28282814.
- ^ a b c d e f g h i j k l m n o p q r s t u Jankovic J (April 2008). "Parkinson's disease: clinical features and diagnosis". Journal of Neurology, Neurosurgery, and Psychiatry. 79 (4): 368–376. doi:10.1136/jnnp.2007.131045. PMID 18344392. Archived from the original on 19 August 2015.
- ^ Winiker & Kertscher 2023, sec. "Introduction". Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License.
- ^ Sveinbjornsdottir S (October 2016). "The clinical symptoms of Parkinson's disease". Journal of Neurochemistry. 139 (S1): 318–324. doi:10.1111/jnc.13691. ISSN 0022-3042. PMID 27401947.
- ^ a b Cooper, Eichhorn & Rodnitzky 2008, pp. 508–512.
- ^ Lees AJ, Hardy J, Revesz T (June 2009). "Parkinson's disease". Lancet. 373 (9680): 2055–2066. doi:10.1016/S0140-6736(09)60492-X. PMID 19524782. S2CID 42608600.
- ^ Fung & Thompson 2007, pp. 504–513.
- ^ O'Sullivan & Schmitz 2007, pp. 856–857.
- ^ Yao SC, Hart AD, Terzella MJ (May 2013). "An evidence-based osteopathic approach to Parkinson disease". Osteopathic Family Physician. 5 (3): 96–101. doi:10.1016/j.osfp.2013.01.003.
- ^ Hallett M, Poewe W (2008). Therapeutics of Parkinson's Disease and Other Movement Disorders. John Wiley & Sons. p. 417. ISBN 978-0-470-71400-3. Archived from the original on 8 September 2017.
- ^ Hoehn MM, Yahr MD (May 1967). "Parkinsonism: onset, progression and mortality". Neurology. 17 (5): 427–442. doi:10.1212/wnl.17.5.427. PMID 6067254.
- ^ Pahwa R, Lyons KE (2003). Handbook of Parkinson's Disease (Third ed.). CRC Press. p. 76. ISBN 978-0-203-91216-4. Archived from the original on 8 September 2017.
- ^ a b Prenger MTM, Madray R, Van Hedger K, Anello M, MacDonald PA (2020). "Social Symptoms of Parkinson's Disease". Parkinsons Dis. 2020: 8846544. doi:10.1155/2020/8846544. PMC 7790585. PMID 33489081.
- ^ a b c d e f g Caballol N, Martí MJ, Tolosa E (September 2007). "Cognitive dysfunction and dementia in Parkinson disease". Movement Disorders. 22 (Suppl 17): S358–S366. doi:10.1002/mds.21677. PMID 18175397. S2CID 3229727.
- ^ a b c Parker KL, Lamichhane D, Caetano MS, Narayanan NS (October 2013). "Executive dysfunction in Parkinson's disease and timing deficits". Frontiers in Integrative Neuroscience. 7: 75. doi:10.3389/fnint.2013.00075. PMC 3813949. PMID 24198770.
- ^ a b c d e f g h i Han JW, Ahn YD, Kim WS, Shin CM, Jeong SJ, Song YS, et al. (November 2018). "Psychiatric Manifestation in Patients with Parkinson's Disease". Journal of Korean Medical Science. 33 (47): e300. doi:10.3346/jkms.2018.33.e300. PMC 6236081. PMID 30450025.
- ^ Ffytche DH, Creese B, Politis M, Chaudhuri KR, Weintraub D, Ballard C, Aarsland D (February 2017). "The psychosis spectrum in Parkinson disease". Nature Reviews. Neurology. 13 (2): 81–95. doi:10.1038/nrneurol.2016.200. PMC 5656278. PMID 28106066.
- ^ Shergill SS, Walker Z, Le Katona C (October 1998). "A preliminary investigation of laterality in Parkinson's disease and susceptibility to psychosis". Journal of Neurology, Neurosurgery, and Psychiatry. 65 (4): 610–611. doi:10.1136/jnnp.65.4.610. PMC 2170290. PMID 9771806.
- ^ Friedman JH (November 2010). "Parkinson's disease psychosis 2010: a review article". Parkinsonism & Related Disorders. 16 (9): 553–560. doi:10.1016/j.parkreldis.2010.05.004. PMID 20538500.
- ^ a b c d Weintraub D, Mamikonyan E (September 2019). "The Neuropsychiatry of Parkinson Disease: A Perfect Storm". The American Journal of Geriatric Psychiatry. 27 (9): 998–1018. doi:10.1016/j.jagp.2019.03.002. PMC 7015280. PMID 31006550.
- ^ a b c Dissanayaka 2021, pp. 139–156.
- ^ a b Goetz CG (2010). "New developments in depression, anxiety, compulsiveness, and hallucinations in Parkinson's disease". Movement Disorders. 25 (S1): S104–S109. doi:10.1002/mds.22636. PMID 20187250. S2CID 35420377.
- ^ Husain M, Roiser JP (August 2018). "Neuroscience of apathy and anhedonia: a transdiagnostic approach". Nature Reviews. Neuroscience. 19 (8): 470–484. doi:10.1038/s41583-018-0029-9. PMID 29946157. S2CID 49428707.
- ^ Turner V, Husain M (2022). "Anhedonia in Neurodegenerative Diseases". Current Topics in Behavioral Neurosciences. 58: 255–277. doi:10.1007/7854_2022_352. ISBN 978-3-031-09682-2. PMID 35435648.
- ^ Noyce AJ, Bestwick JP, Silveira-Moriyama L, et al. (December 2012). "Meta-analysis of early nonmotor features and risk factors for Parkinson disease". Annals of Neurology (Review). 72 (6): 893–901. doi:10.1002/ana.23687. PMC 3556649. PMID 23071076.
- ^ a b c d e f g h i j Barichella M, Cereda E, Pezzoli G (October 2009). "Major nutritional issues in the management of Parkinson's disease". Movement Disorders. 24 (13): 1881–1892. doi:10.1002/mds.22705. hdl:2434/67795. PMID 19691125. S2CID 23528416.
- ^ Warnecke T, Schäfer KH, Claus I, Del Tredici K, Jost WH (March 2022). "Gastrointestinal involvement in Parkinson's disease: pathophysiology, diagnosis, and management". npj Parkinson's Disease. 8 (1): 31. doi:10.1038/s41531-022-00295-x. PMC 8948218. PMID 35332158.
- ^ a b c Skjærbæk C, Knudsen K, Horsager J, Borghammer P (January 2021). "Gastrointestinal Dysfunction in Parkinson's Disease". J Clin Med. 10 (3): 493. doi:10.3390/jcm10030493. PMC 7866791. PMID 33572547.
- ^ Kim YE, Jeon BS (1 January 2014). "Clinical implication of REM sleep behavior disorder in Parkinson's disease". Journal of Parkinson's Disease. 4 (2): 237–244. doi:10.3233/jpd-130293. PMID 24613864.
- ^ Panicker 2021, pp. 1–2.
- ^ a b Toloda 2021, pp. 2.
- ^ a b c d e f g de Lau LM, Breteler MM (June 2006). "Epidemiology of Parkinson's disease". The Lancet. Neurology. 5 (6): 525–535. doi:10.1016/S1474-4422(06)70471-9. PMID 16713924. S2CID 39310242.
- ^ Toloda 2021, pp. 385.
- ^ Barreto GE, Iarkov A, Moran VE (January 2015). "Beneficial effects of nicotine, cotinine and its metabolites as potential agents for Parkinson's disease". Frontiers in Aging Neuroscience. 6: 340. doi:10.3389/fnagi.2014.00340. PMC 4288130. PMID 25620929.
- ^ a b De Miranda 2022, pp. 46.
- ^ "Parkinson's disease - Causes". nhs.uk. 3 October 2018. Retrieved 24 January 2024.
- ^ a b c Lesage S, Brice A (April 2009). "Parkinson's disease: from monogenic forms to genetic susceptibility factors". Human Molecular Genetics. 18 (R1): R48–59. doi:10.1093/hmg/ddp012. PMID 19297401.
- ^ Deng H, Wang P, Jankovic J (March 2018). "The genetics of Parkinson disease". Ageing Research Reviews. 42: 72–85. doi:10.1016/j.arr.2017.12.007. PMID 29288112. S2CID 28246244.
- ^ a b Toloda 2021, pp. 388.
- ^ Blauwendraat 2019, pp. 1.
- ^ Polissidis 2020, pp. 2.
- ^ a b Panicker 2021, pp. 1.
- ^ a b Abugable AA, Morris JL, Palminha NM, et al. (September 2019). "DNA repair and neurological disease: From molecular understanding to the development of diagnostics and model organisms". DNA Repair. 81: 102669. doi:10.1016/j.dnarep.2019.102669. PMID 31331820.
- ^ Blauwendraat 2019, pp. 3.
- ^ a b c Simon DK, Tanner CM, Brundin P (2020). "Parkinson Disease Epidemiology, Pathology, Genetics, and Pathophysiology". Clinical Geriatric Medicine. 36 (1): 1–12. doi:10.1016/j.cger.2019.08.002. PMC 6905381. PMID 31733690. S2CID 201961979.
- ^ Islam 2021, pp. 2–3.
- ^ "Parkinson's Disease". National Institute of Environmental Health Sciences. NIEHS. 3 November 2023. Retrieved 4 December 2023. This article incorporates text from this source, which is in the public domain.
- ^ Islam 2021, pp. 3.
- ^ Islam 2021, pp. 2.
- ^ a b c d e f Simon, Greenberg & Aminoff 2017, p. page number needed.
- ^ Langston JW (6 March 2017). "The MPTP Story". Journal of Parkinson's Disease. 7 (s1): S11–S19. doi:10.3233/JPD-179006. PMC 5345642. PMID 28282815.
- ^ Song L, Xu MB, Zhou XL, Zhang DP, Zhang SL, Zheng GQ (2017). "A Preclinical Systematic Review of Ginsenoside-Rg1 in Experimental Parkinson's Disease". Oxidative Medicine and Cellular Longevity. 2017: 2163053. doi:10.1155/2017/2163053. PMC 5366755. PMID 28386306.
- ^ Chahine LM, Stern MB, Chen-Plotkin A (January 2014). "Blood-based biomarkers for Parkinson's disease". Parkinsonism & Related Disorders. 20 (Suppl 1): S99–103. doi:10.1016/S1353-8020(13)70025-7. PMC 4070332. PMID 24262199.
- ^ Çamcı G, Oğuz S (April 2016). "Association between Parkinson's Disease and Helicobacter Pylori". Journal of Clinical Neurology. 12 (2): 147–150. doi:10.3988/jcn.2016.12.2.147. PMC 4828559. PMID 26932258.
- ^ McGee DJ, Lu XH, Disbrow EA (2018). "Stomaching the Possibility of a Pathogenic Role for Helicobacter pylori in Parkinson's Disease". Journal of Parkinson's Disease. 8 (3): 367–374. doi:10.3233/JPD-181327. PMC 6130334. PMID 29966206.
- ^ Dorsey ER, Zafar M, Lettenberger SE, et al. (2023). "Trichloroethylene: An Invisible Cause of Parkinson's Disease?". J Parkinsons Dis. 13 (2): 203–218. doi:10.3233/JPD-225047. PMC 10041423. PMID 36938742.
- ^ Bjorklund G, Stejskal V, Urbina MA, Dadar M, Chirumbolo S, Mutter J (2018). "Metals and Parkinson's Disease: Mechanisms and Biochemical Processes" (PDF). Curr Med Chem. 25 (19): 2198–2214. doi:10.2174/0929867325666171129124616. PMID 29189118. S2CID 4648656.
- ^ Delic V, Beck KD, Pang KC, Citron BA (2020). "Biological links between traumatic brain injury and Parkinson's disease". Acta Neuropathologica Communications. 8 (1): 45. doi:10.1186/s40478-020-00924-7. PMC 7137235. PMID 32264976.
- ^ Ascherio 2016, pp. 1260.
- ^ a b c Davie CA (2008). "A review of Parkinson's disease". British Medical Bulletin. 86 (1): 109–127. doi:10.1093/bmb/ldn013. PMID 18398010.
- ^ Villar-Piqué A, Lopes da Fonseca T, Outeiro TF (October 2016). "Structure, function and toxicity of alpha-synuclein: the Bermuda triangle in synucleinopathies". Journal of Neurochemistry. 139 (Suppl 1): 240–255. doi:10.1111/jnc.13249. PMID 26190401. S2CID 11420411.
- ^ Burré J, Sharma M, Südhof TC (March 2018). "Cell Biology and Pathophysiology of α-Synuclein". Cold Spring Harbor Perspectives in Medicine. 8 (3): a024091. doi:10.1101/cshperspect.a024091. PMC 5519445. PMID 28108534.
- ^ a b Dickson 2007, pp. 271–283.
- ^ a b c d e f g h i j Armstrong MJ, Okun MS (February 2020). "Diagnosis and Treatment of Parkinson Disease: A Review". JAMA. 323 (6): 548–560. doi:10.1001/jama.2019.22360. PMID 32044947. S2CID 211079287.
- ^ a b c d e f Obeso JA, Rodríguez-Oroz MC, Benitez-Temino B, Blesa FJ, Guridi J, Marin C, Rodriguez M (2008). "Functional organization of the basal ganglia: therapeutic implications for Parkinson's disease". Movement Disorders. 23 (Suppl 3): S548–S559. doi:10.1002/mds.22062. PMID 18781672. S2CID 13186083.
- ^ a b c Schulz-Schaeffer WJ (August 2010). "The synaptic pathology of alpha-synuclein aggregation in dementia with Lewy bodies, Parkinson's disease and Parkinson's disease dementia". Acta Neuropathologica. 120 (2): 131–143. doi:10.1007/s00401-010-0711-0. PMC 2892607. PMID 20563819.
- ^ a b c d e Obeso JA, Rodriguez-Oroz MC, Goetz CG, et al. (June 2010). "Missing pieces in the Parkinson's disease puzzle". Nature Medicine. 16 (6): 653–661. doi:10.1038/nm.2165. PMID 20495568. S2CID 3146438.
- ^ Hirsch EC (December 2009). "Iron transport in Parkinson's disease". Parkinsonism & Related Disorders. 15 (Suppl 3): S209–S211. doi:10.1016/S1353-8020(09)70816-8. PMID 20082992.
- ^ Li X, Sundquist J, Sundquist K (23 December 2011). "Subsequent Risks of Parkinson Disease in Patients with Autoimmune and Related Disorders: A Nationwide Epidemiological Study from Sweden". Neurodegenerative Diseases. 10 (1–4): 277–284. doi:10.1159/000333222. ISSN 1660-2854. PMID 22205172. S2CID 39874367.
- ^ Lai SW, Lin CH, Lin HF, Lin CL, Lin CC, Liao KF (February 2017). "Herpes zoster correlates with increased risk of Parkinson's disease in older people: A population-based cohort study in Taiwan". Medicine (Baltimore). 96 (7): e6075. doi:10.1097/MD.0000000000006075. PMC 5319504. PMID 28207515.
- ^ Tan EK, Chao YX, West A, Chan LL, Poewe W, Jankovic J (June 2020). "Parkinson disease and the immune system - associations, mechanisms and therapeutics". Nat Rev Neurol. 16 (6): 303–318. doi:10.1038/s41582-020-0344-4. ISSN 1759-4758. PMID 32332985. S2CID 216111568.
- ^ Raj T, Rothamel K, Mostafavi S, Ye C, Lee MN, Replogle JM, et al. (May 2014). "Polarization of the effects of autoimmune and neurodegenerative risk alleles in leukocytes". Science. 344 (6183): 519–23. Bibcode:2014Sci...344..519R. doi:10.1126/science.1249547. PMC 4910825. PMID 24786080.
- ^ a b Du G, Dong W, Yang Q, Yu X, Ma J, Gu W, Huang Y (2020). "Altered Gut Microbiota Related to Inflammatory Responses in Patients With Huntington's Disease". Front Immunol. 11: 603594. doi:10.3389/fimmu.2020.603594. ISSN 1664-3224. PMC 7933529. PMID 33679692.
- ^ a b Gamborg M, Hvid LG, Dalgas U, Langeskov-Christensen M (3 October 2021). "Review for "Parkinson's disease and intensive exercise therapy — An updated systematic review and meta-analysis"". Acta Neurologica Scandinavica. doi:10.1111/ane.13579/v1/review2. ISSN 1600-0404.
- ^ [non-primary source needed]Kim S, Kwon SH, Kam TI, Panicker N, Karuppagounder SS, Lee S, et al. (August 2019). "Transneuronal Propagation of Pathologic α-Synuclein from the Gut to the Brain Models Parkinson's Disease". Neuron. 103 (4): 627–641.e7. doi:10.1016/j.neuron.2019.05.035. PMC 6706297. PMID 31255487.
- ^ a b The National Collaborating Centre for Chronic Conditions, ed. (2006). "Diagnosing Parkinson's Disease". Parkinson's Disease. London: Royal College of Physicians. pp. 29–47. ISBN 978-1-86016-283-1. Archived from the original on 24 September 2010.
- ^ Poewe W, Wenning G (November 2002). "The differential diagnosis of Parkinson's disease". European Journal of Neurology. 9 (Suppl 3): 23–30. doi:10.1046/j.1468-1331.9.s3.3.x. PMID 12464118.
- ^ Gibb WR, Lees AJ (June 1988). "The relevance of the Lewy body to the pathogenesis of idiopathic Parkinson's disease". Journal of Neurology, Neurosurgery, and Psychiatry. 51 (6): 745–752. doi:10.1136/jnnp.51.6.745. PMC 1033142. PMID 2841426.
- ^ Mustafa HI, Fessel JP, Barwise J, Shannon JR, Raj SR, Diedrich A, et al. (January 2012). "Dysautonomia: perioperative implications". Anesthesiology. 116 (1): 205–215. doi:10.1097/ALN.0b013e31823db712. PMC 3296831. PMID 22143168.
- ^ Rizzo G, Copetti M, Arcuti S, Martino D, Fontana A, Logroscino G (February 2016). "Accuracy of clinical diagnosis of Parkinson disease: A systematic review and meta-analysis". Neurology. 86 (6): 566–576. doi:10.1212/WNL.0000000000002350. PMID 26764028. S2CID 207110404.
- ^ a b c d Brooks DJ (April 2010). "Imaging approaches to Parkinson disease". Journal of Nuclear Medicine. 51 (4): 596–609. doi:10.2967/jnumed.108.059998. PMID 20351351.
- ^ Schwarz ST, Afzal M, Morgan PS, Bajaj N, Gowland PA, Auer DP (2014). "The 'swallow tail' appearance of the healthy nigrosome – a new accurate test of Parkinson's disease: a case-control and retrospective cross-sectional MRI study at 3T". PLOS ONE. 9 (4): e93814. Bibcode:2014PLoSO...993814S. doi:10.1371/journal.pone.0093814. PMC 3977922. PMID 24710392.
- ^ Mahlknecht P, Krismer F, Poewe W, Seppi K (April 2017). "Meta-analysis of dorsolateral nigral hyperintensity on magnetic resonance imaging as a marker for Parkinson's disease". Movement Disorders. 32 (4): 619–623. doi:10.1002/mds.26932. PMID 28151553. S2CID 7730034.
- ^ Cho SJ, Bae YJ, Kim JM, et al. (September 2020). "Diagnostic performance of neuromelanin-sensitive magnetic resonance imaging for patients with Parkinson's disease and factor analysis for its heterogeneity: a systematic review and meta-analysis". European Radiology. 30 (10): 1268–1280. doi:10.1007/s00330-020-07240-7. PMID 32886201. S2CID 221478854.
- ^ Boonstra JT, Michielse S, Temel Y, Hoogland G, Jahanshahi A (February 2021). "Neuroimaging Detectable Differences between Parkinson's Disease Motor Subtypes: A Systematic Review". Movement Disorders Clinical Practice. 8 (2): 175–192. doi:10.1002/mdc3.13107. PMC 7853198. PMID 33553487.
- ^ Suwijn SR, van Boheemen CJ, de Haan RJ, Tissingh G, Booij J, de Bie RM (2015). "The diagnostic accuracy of dopamine transporter SPECT imaging to detect nigrostriatal cell loss in patients with Parkinson's disease or clinically uncertain parkinsonism: a systematic review". EJNMMI Research. 5: 12. doi:10.1186/s13550-015-0087-1. PMC 4385258. PMID 25853018.
- ^ "DaTSCAN Approval Letter" (PDF). FDA.gov. Food and Drug Administration. Retrieved 22 March 2019.
- ^ a b c d e Greenland & Barker 2018.
- ^ McCann H, Stevens CH, Cartwright H, Halliday GM (January 2014). "α-Synucleinopathy phenotypes". Parkinsonism & Related Disorders. 20 (Suppl 1): S62–S67. doi:10.1016/S1353-8020(13)70017-8. hdl:1959.4/53593. PMID 24262191.
- ^ Ganguly J, Jog M (5 November 2020). "Tauopathy and Movement Disorders-Unveiling the Chameleons and Mimics". Frontiers in Neurology. 11: 599384. doi:10.3389/fneur.2020.599384. PMC 7674803. PMID 33250855.
- ^ Gupta D, Kuruvilla A (December 2011). "Vascular parkinsonism: what makes it different?". Postgraduate Medical Journal. 87 (1034): 829–836. doi:10.1136/postgradmedj-2011-130051. PMID 22121251. S2CID 29227069.
- ^ Miguel-Puga A, Villafuerte G, Salas-Pacheco J, Arias-Carrión O (22 September 2017). "Therapeutic Interventions for Vascular Parkinsonism: A Systematic Review and Meta-analysis". Frontiers in Neurology. 8: 481. doi:10.3389/fneur.2017.00481. PMC 5614922. PMID 29018399.
- ^ Levin J, Kurz A, Arzberger T, Giese A, Höglinger GU (February 2016). "The Differential Diagnosis and Treatment of Atypical Parkinsonism". Deutsches Ärzteblatt International. 113 (5): 61–69. doi:10.3238/arztebl.2016.0061. PMC 4782269. PMID 26900156.
- ^ a b Ahlskog JE (July 2011). "Does vigorous exercise have a neuroprotective effect in Parkinson disease?". Neurology. 77 (3): 288–294. doi:10.1212/wnl.0b013e318225ab66. PMC 3136051. PMID 21768599.
- ^ Ascherio A, Schwarzschild MA (November 2016). "The epidemiology of Parkinson's disease: risk factors and prevention". The Lancet Neurology. 15 (12): 1257–1272. doi:10.1016/S1474-4422(16)30230-7. PMID 27751556. S2CID 19994300.
- ^ Costa J, Lunet N, Santos C, Santos J, Vaz-Carneiro A (2010). "Caffeine exposure and the risk of Parkinson's disease: a systematic review and meta-analysis of observational studies". Journal of Alzheimer's Disease. 20 (Suppl 1): S221–238. doi:10.3233/JAD-2010-091525. PMID 20182023.
- ^ Gagne JJ, Power MC (March 2010). "Anti-inflammatory drugs and risk of Parkinson disease: a meta-analysis". Neurology. 74 (12): 995–1002. doi:10.1212/WNL.0b013e3181d5a4a3. PMC 2848103. PMID 20308684.
- ^ Elkouzi A, Vedam-Mai V, Eisinger RS, Okun MS (April 2019). "Emerging therapies in Parkinson disease – repurposed drugs and new approaches". Nat Rev Neurol. 15 (4): 204–223. doi:10.1038/s41582-019-0155-7. PMC 7758837. PMID 30867588.
- ^ Connolly BS, Lang AE (30 April 2014). "Pharmacological treatment of Parkinson disease: a review". JAMA. 311 (16): 1670–1683. doi:10.1001/jama.2014.3654. PMID 24756517. S2CID 205058847.
- ^ Olanow CW, Stocchi F, Lang AE (2011). "The non-motor and non-dopaminergic features of PD". Parkinson's Disease: Non-Motor and Non-Dopaminergic Features. Wiley-Blackwell. ISBN 978-1-4051-9185-2. OCLC 743205140.
- ^ a b c d e f g h i j k l m n o p The National Collaborating Centre for Chronic Conditions, ed. (2006). "Symptomatic pharmacological therapy in Parkinson's disease". Parkinson's Disease (Technical report). London: Royal College of Physicians. pp. 59–100. ISBN 978-1-86016-283-1. Archived from the original on 24 September 2010. Retrieved 14 March 2023.
- ^ Zhang J, Tan LC (2016). "Revisiting the Medical Management of Parkinson's Disease: Levodopa versus Dopamine Agonist". Current Neuropharmacology. 14 (4): 356–363. doi:10.2174/1570159X14666151208114634. PMC 4876591. PMID 26644151.
- ^ Moosa S, Martínez-Fernández R, Elias WJ, Del Alamo M, Eisenberg HM, Fishman PS (September 2019). "The role of high-intensity focused ultrasound as a symptomatic treatment for Parkinson's disease". Mov Disord. 34 (9): 1243–1251. doi:10.1002/mds.27779. ISSN 0885-3185. PMID 31291491. S2CID 195879250.
- ^ a b Pedrosa DJ, Timmermann L (2013). "Review: management of Parkinson's disease". Neuropsychiatric Disease and Treatment (Review). 9: 321–340. doi:10.2147/NDT.S32302. PMC 3592512. PMID 23487540.
- ^ The National Collaborating Centre for Chronic Conditions, ed. (2006). "Palliative care in Parkinson's disease". Parkinson's Disease. London: Royal College of Physicians. pp. 147–151. ISBN 978-1-86016-283-1. Archived from the original on 24 September 2010.
- ^ Orgeta V, McDonald KR, Poliakoff E, Hindle JV, Clare L, Leroi I (February 2020). "Cognitive training interventions for dementia and mild cognitive impairment in Parkinson's disease". The Cochrane Database of Systematic Reviews. 2020 (2): CD011961. doi:10.1002/14651858.cd011961.pub2. PMC 7043362. PMID 32101639.
- ^ a b c d e f g h i "Recommendations Parkinson's disease in adults Guidance NICE". NICE. 2006. Retrieved 16 March 2023.
- ^ Maria N (2017). Levodopa pharmacokinetics – from stomach to brain: A study on patients with Parkinson's disease. Linköping: Linköping University Electronic Press. p. 10. ISBN 978-91-7685-557-7. OCLC 993068595.
- ^ Oertel WH (13 March 2017). "Recent advances in treating Parkinson's disease". F1000Research (Review). 6: 260. doi:10.12688/f1000research.10100.1. PMC 5357034. PMID 28357055.
- ^ Binde et al. 2018, p. missing.
- ^ Aquino CC, Fox SH (January 2015). "Clinical spectrum of levodopa-induced complications". Movement Disorders. 30 (1): 80–89. doi:10.1002/mds.26125. PMID 25488260. S2CID 22301199.
- ^ Yan Z, Wang W, Tao X, Cheng W, Zuo G, Chen Z (30 July 2022). "High-dose versus low-dose inhaled levodopa (CVT-301) in patients with Parkinson disease for the treatment of OFF episodes: a meta-analysis of randomized controlled trials". Neurological Sciences. 43 (11): 6233–6241. doi:10.1007/s10072-022-06298-z. PMID 35907110. S2CID 251162631. Retrieved 14 March 2023.
- ^ Paik J (June 2020). "Levodopa Inhalation Powder: A Review in Parkinson's Disease". Drugs. 80 (8): 821–828. doi:10.1007/s40265-020-01307-x. PMID 32319076. S2CID 216033034.
- ^ Tambasco N, Romoli M, Calabresi P (2018). "Levodopa in Parkinson's Disease: Current Status and Future Developments". Current Neuropharmacology. 16 (8): 1239–1252. doi:10.2174/1570159X15666170510143821. PMC 6187751. PMID 28494719.
- ^ a b c Akhtar MJ, Yar MS, Grover G, Nath R (January 2020). "Neurological and psychiatric management using COMT inhibitors: A review". Bioorganic Chemistry. 94: 103418. doi:10.1016/j.bioorg.2019.103418. PMID 31708229.
- ^ "Tasmar 100 mg Tablets – Summary of Product Characteristics (SmPC) – (emc)". www.medicines.org.uk. Archived from the original on 6 August 2020. Retrieved 7 January 2021.
- ^ Scott LJ (September 2016). "Opicapone: A Review in Parkinson's Disease". Drugs. 76 (13): 1293–1300. doi:10.1007/s40265-016-0623-y. PMID 27498199. S2CID 5787752.
- ^ Watkins P (2000). "COMT inhibitors and liver toxicity". Neurology. 55 (11 Suppl 4): S51–52, discussion S53–56. PMID 11147510.
- ^ "Comtess 200 mg film-coated Tablets – Summary of Product Characteristics (SmPC) – (emc)". www.medicines.org.uk. Retrieved 7 January 2021.
- ^ "Stalevo 150 mg/37.5 mg/200 mg Film-coated Tablets – Summary of Product Characteristics (SmPC) – (emc)". www.medicines.org.uk. Retrieved 7 January 2021.
- ^ "Ongentys 50 mg hard capsules – Summary of Product Characteristics (SmPC) – (emc)". www.medicines.org.uk. Retrieved 7 January 2021.
- ^ Goldenberg MM (October 2008). "Medical management of Parkinson's disease". P & T. 33 (10): 590–606. PMC 2730785. PMID 19750042.
- ^ Ceravolo R, Frosini D, Rossi C, Bonuccelli U (December 2009). "Impulse control disorders in Parkinson's disease: definition, epidemiology, risk factors, neurobiology and management". Parkinsonism & Related Disorders. 15 (Suppl 4): S111–S115. doi:10.1016/S1353-8020(09)70847-8. PMID 20123548.
- ^ Tolosa & Katzenschlager 2007, pp. 110–145.
- ^ Binde et al. 2018, p. 1924.
- ^ a b c Alborghetti M, Nicoletti F (2019). "Different Generations of Type-B Monoamine Oxidase Inhibitors in Parkinson's Disease: From Bench to Bedside". Current Neuropharmacology. 17 (9): 861–873. doi:10.2174/1570159X16666180830100754. PMC 7052841. PMID 30160213.
- ^ Ives NJ, Stowe RL, Marro J, Counsell C, Macleod A, Clarke CE, et al. (September 2004). "Monoamine oxidase type B inhibitors in early Parkinson's disease: meta-analysis of 17 randomised trials involving 3525 patients". BMJ. 329 (7466): 593. doi:10.1136/bmj.38184.606169.AE. PMC 516655. PMID 15310558.
- ^ Crosby N, Deane KH, Clarke CE (2003). "Amantadine in Parkinson's disease". The Cochrane Database of Systematic Reviews. 2010 (1): CD003468. doi:10.1002/14651858.CD003468. PMC 8715353. PMID 12535476.
- ^ a b The National Collaborating Centre for Chronic Conditions, ed. (2006). "Non-motor features of Parkinson's disease". Parkinson's Disease. London: Royal College of Physicians. pp. 113–133. ISBN 978-1-86016-283-1. Archived from the original on 24 September 2010.
- ^ Hasnain M, Vieweg WV, Baron MS, Beatty-Brooks M, Fernandez A, Pandurangi AK (July 2009). "Pharmacological management of psychosis in elderly patients with parkinsonism". The American Journal of Medicine. 122 (7): 614–622. doi:10.1016/j.amjmed.2009.01.025. PMID 19559160.
- ^ "FDA approves first drug to treat hallucinations and delusions associated with Parkinson's disease". www.fda.gov (Press release). 29 April 2016. Retrieved 12 October 2018.
- ^ a b Elbers RG, Verhoef J, van Wegen EE, Berendse HW, Kwakkel G (October 2015). "Interventions for fatigue in Parkinson's disease". The Cochrane Database of Systematic Reviews (Review). 2015 (10): CD010925. doi:10.1002/14651858.CD010925.pub2. PMC 9240814. PMID 26447539.
- ^ a b c d The National Collaborating Centre for Chronic Conditions, ed. (2006). "Surgery for Parkinson's disease". Parkinson's Disease. London: Royal College of Physicians. pp. 101–111. ISBN 978-1-86016-283-1. Archived from the original on 24 September 2010.
- ^ Bronstein JM, Tagliati M, Alterman RL, et al. (February 2011). "Deep brain stimulation for Parkinson disease: an expert consensus and review of key issues". Archives of Neurology. 68 (2): 165. doi:10.1001/archneurol.2010.260. PMC 4523130. PMID 20937936.
- ^ a b Dallapiazza et al. 2018.
- ^ Ernst M, Folkerts AK, Gollan R, Lieker E, Caro-Valenzuela J, Adams A, Cryns N, Monsef I, Dresen A, Roheger M, Eggers C, Skoetz N, Kalbe E (5 January 2023). Cochrane Movement Disorders Group (ed.). "Physical exercise for people with Parkinson's disease: a systematic review and network meta-analysis". Cochrane Database of Systematic Reviews. 2023 (5): CD013856. doi:10.1002/14651858.CD013856.pub2. PMC 9815433. PMID 36602886.
- ^ a b c d e The National Collaborating Centre for Chronic Conditions, ed. (2006). "Other key interventions". Parkinson's Disease. London: Royal College of Physicians. pp. 135–146. ISBN 978-1-86016-283-1. Archived from the original on 24 September 2010.
- ^ a b Goodwin VA, Richards SH, Taylor RS, Taylor AH, Campbell JL (April 2008). "The effectiveness of exercise interventions for people with Parkinson's disease: a systematic review and meta-analysis". Movement Disorders. 23 (5): 631–640. doi:10.1002/mds.21922. hdl:10871/17451. PMID 18181210. S2CID 3808899.
- ^ Dereli EE, Yaliman A (April 2010). "Comparison of the effects of a physiotherapist-supervised exercise programme and a self-supervised exercise programme on quality of life in patients with Parkinson's disease". Clinical Rehabilitation. 24 (4): 352–362. doi:10.1177/0269215509358933. PMID 20360152. S2CID 10947269.
- ^ Jin X, Wang L, Liu S, Zhu L, Loprinzi PD, Fan X (December 2019). "The Impact of Mind-body Exercises on Motor Function, Depressive Symptoms, and Quality of Life in Parkinson's Disease: A Systematic Review and Meta-analysis". International Journal of Environmental Research and Public Health. 17 (1): 31. doi:10.3390/ijerph17010031. PMC 6981975. PMID 31861456.
- ^ O'Sullivan & Schmitz 2007, pp. 873, 876.
- ^ O'Sullivan & Schmitz 2007, p. 879.
- ^ O'Sullivan & Schmitz 2007, p. 877.
- ^ O'Sullivan & Schmitz 2007, p. 880.
- ^ Ramazzina I, Bernazzoli B, Costantino C (31 March 2017). "Systematic review on strength training in Parkinson's disease: an unsolved question". Clinical Interventions in Aging. 12: 619–628. doi:10.2147/CIA.S131903. PMC 5384725. PMID 28408811.
- ^ Fox CM, Ramig LO, Ciucci MR, Sapir S, McFarland DH, Farley BG (November 2006). "The science and practice of LSVT/LOUD: neural plasticity-principled approach to treating individuals with Parkinson disease and other neurological disorders". Seminars in Speech and Language. 27 (4): 283–299. doi:10.1055/s-2006-955118. PMID 17117354. S2CID 260320960.
- ^ Dixon L, Duncan D, Johnson P, et al. (July 2007). "Occupational therapy for patients with Parkinson's disease". The Cochrane Database of Systematic Reviews. 2007 (3): CD002813. doi:10.1002/14651858.CD002813.pub2. PMC 6991932. PMID 17636709.
- ^ Ferrell B, Connor SR, Cordes A, et al. (June 2007). "The national agenda for quality palliative care: the National Consensus Project and the National Quality Forum". Journal of Pain and Symptom Management. 33 (6): 737–744. doi:10.1016/j.jpainsymman.2007.02.024. PMID 17531914.
- ^ a b Lorenzl S, Nübling G, Perrar KM, Voltz R (2013). "Palliative treatment of chronic neurologic disorders". Ethical and Legal Issues in Neurology. Handbook of Clinical Neurology. Vol. 118. Elsevier. pp. 133–139. doi:10.1016/B978-0-444-53501-6.00010-X. ISBN 978-0-444-53501-6. PMID 24182372.
- ^ a b Ghoche R (December 2012). "The conceptual framework of palliative care applied to advanced Parkinson's disease". Parkinsonism & Related Disorders. 18 (Suppl 3): S2–5. doi:10.1016/j.parkreldis.2012.06.012. PMID 22771241.
- ^ a b c Wilcox SK (January 2010). "Extending palliative care to patients with Parkinson's disease". British Journal of Hospital Medicine. 71 (1): 26–30. doi:10.12968/hmed.2010.71.1.45969. PMID 20081638.
- ^ Moens K, Higginson IJ, Harding R (October 2014). "Are there differences in the prevalence of palliative care-related problems in people living with advanced cancer and eight non-cancer conditions? A systematic review". Journal of Pain and Symptom Management. 48 (4): 660–677. doi:10.1016/j.jpainsymman.2013.11.009. PMID 24801658.
- ^ Casey G (August 2013). "Parkinson's disease: a long and difficult journey". Nursing New Zealand. 19 (7): 20–24. PMID 24195263.
- ^ a b c d Poewe W (December 2006). "The natural history of Parkinson's disease". Journal of Neurology. 253 (Suppl 7): vii2–vii6. doi:10.1007/s00415-006-7002-7. PMID 17131223. S2CID 35082340.
- ^ a b Feigin VL, Nichols E, Alam T, et al. (May 2019). "Global, regional, and national burden of neurological disorders, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016". Lancet Neurol. 18 (5): 459–480. doi:10.1016/S1474-4422(18)30499-X. PMC 6459001. PMID 30879893.
- ^ Gomperts SN (April 2016). "Lewy Body Dementias: Dementia With Lewy Bodies and Parkinson Disease Dementia". Continuum (Minneap Minn) (Review). 22 (2 Dementia): 435–463. doi:10.1212/CON.0000000000000309. PMC 5390937. PMID 27042903.
- ^ Binde et al. 2018, p. 1989.
- ^ Li G, Ma J, Cui S, He Y, Xiao Q, Liu J, Chen S (31 July 2019). "Parkinson's disease in China: a forty-year growing track of bedside work". Translational Neurodegeneration. 8 (1): 22. doi:10.1186/s40035-019-0162-z. PMC 6668186. PMID 31384434.
- ^ Dorsey ER, Sherer T, Okun MS, Bloem BR (2018). "The Emerging Evidence of the Parkinson Pandemic". Journal of Parkinson's Disease. 8 (s1): S3–S8. doi:10.3233/JPD-181474. PMC 6311367. PMID 30584159.
- ^ Garcia-Ptacek S, Kramberger MG (September 2016). "Parkinson Disease and Dementia". Journal of Geriatric Psychiatry and Neurology. 29 (5): 261–270. doi:10.1177/0891988716654985. PMID 27502301. S2CID 21279235.
- ^ a b "Art and Medicine Bibliography, Paul de Saint-Léger". Art and Medicine. Retrieved 29 April 2023.
- ^ a b Lewis PA, Plun-Favreau H, Rowley M, Spillane J (March 2020). "Pierre D. and the First Photographs of Parkinson's Disease". Movement Disorders. 35 (3): 389–391. doi:10.1002/MDS.27965. PMC 7155099. PMID 31975439.
- ^ a b c García Ruiz PJ (December 2004). "Prehistoria de la enfermedad de Parkinson" [Prehistory of Parkinson's disease]. Neurologia (in Spanish). 19 (10): 735–737. PMID 15568171.. The article mistakenly refers to Job 34:19 instead of Job 33:19.
- ^ a b Lanska DJ (2009). "Chapter 33 the history of movement disorders". History of Neurology. Handbook of Clinical Neurology. Vol. 95. Elsevier. pp. 501–546. doi:10.1016/S0072-9752(08)02133-7. ISBN 978-0-444-52009-8. PMID 19892136.
- ^ Koehler PJ, Keyser A (September 1997). "Tremor in Latin texts of Dutch physicians: 16th–18th centuries". Movement Disorders. 12 (5): 798–806. doi:10.1002/mds.870120531. PMID 9380070. S2CID 310819.
- ^ Pápai Páriz F (1690). Pax corporis, az az az emberi testnek belső nyavalyáinak okairól, fészkeiről s azoknak orvoslásának módgyáról való tracta., ("Pax corporis, i.e. a teaching of the causes, cources and the methods of treatment of the internal diseases of the human body"). Némethi Mihály, Kolozsvár, Hungarian.
- ^ Bereczki D (2010). "The description of all four cardinal signs of Parkinson's disease in a Hungarian medical text published in 1690". Parkinsonism Relat Disord. 16 (4): 290–293. doi:10.1016/j.parkreldis.2009.11.006. PMID 19948422.
- ^ Bereczki D (2010). "The historically corect name of Parkinson's disease could be Papai-Parkinson's disease". European Journal of Neurology. 16 (Suppl 3): 327. doi:10.1016/j.parkreldis.2009.11.006. PMID 19948422.
- ^ Bereczki D, Balla A, Pelok B, Szatmari S (2018). "International recognition of Ferenc Papai Pariz as one of the first descriptors of all 4 cardinal signs of Parkinson's disease". Orvostudományi Értesítő. 91 (1): 60–66. doi:10.2478/orvtudert-2018-0006.
- ^ a b c d e f g Lees AJ (September 2007). "Unresolved issues relating to the shaking palsy on the celebration of James Parkinson's 250th birthday". Movement Disorders. 22 (Suppl 17): S327–S334. doi:10.1002/mds.21684. PMID 18175393. S2CID 9471754.
- ^ Parkinson J (1817). An Essay on the Shaking Palsy. London: Whittingham and Roland for Sherwood, Neely, and Jones. Archived from the original on 24 September 2015.
- ^ Louis ED (November 1997). "The shaking palsy, the first forty-five years: a journey through the British literature". Movement Disorders. 12 (6): 1068–1072. doi:10.1002/mds.870120638. PMID 9399240. S2CID 34630080.
- ^ a b c Fahn S (2008). "The history of dopamine and levodopa in the treatment of Parkinson's disease". Movement Disorders. 23 (Suppl 3): S497–508. doi:10.1002/mds.22028. PMID 18781671. S2CID 45572523.
- ^ Guridi J, Lozano AM (November 1997). "A brief history of pallidotomy". Neurosurgery. 41 (5): 1169–1180, discussion 1180–1183. doi:10.1097/00006123-199711000-00029. PMID 9361073.
- ^ Hornykiewicz O (2002). "L-DOPA: from a biologically inactive amino acid to a successful therapeutic agent". Amino Acids. 23 (1–3): 65–70. doi:10.1007/s00726-001-0111-9. PMID 12373520. S2CID 25117208.
- ^ Coffey RJ (March 2009). "Deep brain stimulation devices: a brief technical history and review". Artificial Organs. 33 (3): 208–220. doi:10.1111/j.1525-1594.2008.00620.x. PMID 18684199.
- ^ "UF neurologists create a new image of Parkinson disease". McKnight Brain Institute, University of Florida. 27 July 2020. Retrieved 17 November 2023.
- ^ Armstrong MJ, Okun MS (November 2020). "Time for a New Image of Parkinson Disease". JAMA Neurol. 77 (11): 1345–1346. doi:10.1001/jamaneurol.2020.2412. PMID 32716484. S2CID 220796667.
- ^ a b Crooks S, Carter G, Wilson CB, Wynne L, Stark P, Doumas M, Rodger M, O'Shea E, Mitchell G (2023). "Exploring public perceptions and awareness of Parkinson's disease: A scoping review". PLOS ONE. 18 (9): e0291357. Bibcode:2023PLoSO..1891357C. doi:10.1371/journal.pone.0291357. PMC 10503766. PMID 37713383.
- ^ a b c Findley LJ (September 2007). "The economic impact of Parkinson's disease". Parkinsonism & Related Disorders. 13 (Suppl): S8–S12. doi:10.1016/j.parkreldis.2007.06.003. PMID 17702630.
- ^ Yang W, Hamilton JL, Kopil C, et al. (2020). "Current and projected future economic burden of Parkinson's disease in the U.S". npj Parkinson's Disease. 6: 15. doi:10.1038/s41531-020-0117-1. PMC 7347582. PMID 32665974. Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License.
- ^ "Parkinson's – 'the shaking palsy'". GlaxoSmithKline. 1 April 2009. Archived from the original on 14 May 2011.
- ^ "National Parkinson Foundation – Mission". Archived from the original on 21 December 2010. Retrieved 28 March 2011.
- ^ "Education: Joy in Giving". Time. 18 January 1960. Archived from the original on 20 February 2011. Retrieved 2 April 2011.
- ^ "About PDF". Parkinson's Disease Foundation. Archived from the original on 15 May 2011. Retrieved 24 July 2016.
- ^ "American Parkinson Disease Association: Home". American Parkinson Disease Association. Archived from the original on 10 May 2012. Retrieved 9 August 2010.
- ^ "About EPDA". European Parkinson's Disease Association. 2010. Archived from the original on 15 August 2010. Retrieved 9 August 2010.
- ^ "Notable Figures with Parkinson's". Parkinson's Foundation. Retrieved 22 November 2023.
- ^ "Michael's Story". The Michael J. Fox Foundation for Parkinson's Research. Retrieved 7 May 2023.
- ^ Davis P (3 May 2007). "Michael J. Fox". The Time 100. New York: Time. Archived from the original on 25 April 2011. Retrieved 2 April 2011.
- ^ Brockes E (11 April 2009). "'It's the gift that keeps on taking'". The Guardian. London. Archived from the original on 8 October 2013. Retrieved 25 October 2010.
- ^ Burleson N, Breen K (9 November 2023). "Michael J. Fox talks funding breakthrough research for Parkinson's disease". CBS News. Retrieved 23 November 2023.
- ^ Brey RL (April 2006). "Muhammad Ali's Message: Keep Moving Forward". Neurology Now. 2 (2): 8. doi:10.1097/01222928-200602020-00003. Archived from the original on 27 September 2011. Retrieved 22 August 2020.
- ^ Matthews W (April 2006). "Ali's Fighting Spirit". Neurology Now. 2 (2): 10–23. doi:10.1097/01222928-200602020-00004. S2CID 181104230.
- ^ Tauber P (17 July 1988). "Ali: Still Magic". The New York Times. Archived from the original on 17 November 2016. Retrieved 2 April 2011.
- ^ Macur J (26 March 2008). "For the Phinney Family, a Dream and a Challenge". The New York Times. Archived from the original on 6 November 2014. Retrieved 25 May 2013.
About 1.5 million Americans have received a diagnosis of Parkinson's disease, but only 5 to 10 percent learn of it before age 40, according to the National Parkinson Foundation. Davis Phinney was among the few.
- ^ "Who We Are". Davis Phinney Foundation. Archived from the original on 11 January 2012. Retrieved 18 January 2012.
- ^ Jones JM (December 2004). "Great shakes: famous people with Parkinson disease". South Med J. 97 (12): 1186–9. doi:10.1097/01.SMJ.0000145284.79746.B3. PMID 15646755. S2CID 44340931.
- ^ Mufson S (20 February 1997). "China's Deng Xiaoping Is Dead at 92". The Washington Post. Retrieved 30 November 2023.
- ^ "Parkinson's experts sent to help Deng". South China Morning Post. Hong Kong. 26 January 1995. Archived from the original on 30 November 2023. Retrieved 30 November 2023.
- ^ a b Gallman S (4 November 2015). "Robin Williams' widow speaks: Depression didn't kill my husband". CNN. Archived from the original on 4 November 2015. Retrieved 6 April 2018.
- ^ Williams SS (September 2016). "The terrorist inside my husband's brain". Neurology. 87 (13): 1308–1311. doi:10.1212/WNL.0000000000003162. PMID 27672165.
- ^ Robbins R (30 September 2016). "How Lewy body dementia gripped Robin Williams". Scientific American. Retrieved 9 April 2018.
- ^ "LBDA Clarifies Autopsy Report on Comedian, Robin Williams". Lewy Body Dementia Association. 10 November 2014. Archived from the original on 12 August 2020. Retrieved 19 April 2018.
- ^ McKeith IG (6 November 2015). "Robin Williams had dementia with Lewy bodies – so, what is it and why has it been eclipsed by Alzheimer's?". The Conversation. Archived from the original on 4 November 2016. Retrieved 6 April 2018.
- ^ Mari Z, Mestre TA (2022). "The Disease Modification Conundrum in Parkinson's Disease: Failures and Hopes". Frontiers in Aging Neuroscience. 14: 810860. doi:10.3389/fnagi.2022.810860. PMC 8920063. PMID 35296034.
- ^ McFarthing K, Rafaloff G, Baptista M, Mursaleen L, Fuest R, Wyse RK, Stott SR (24 May 2022). "Parkinson's Disease Drug Therapies in the Clinical Trial Pipeline: 2022 Update". Journal of Parkinson's Disease. 12 (4): 1073–1082. doi:10.3233/JPD-229002. PMC 9198738. PMID 35527571.
- ^ a b Poewe W, Seppi K, Tanner CM, Halliday GM, Brundin P, Volkmann J, et al. (March 2017). "Parkinson disease". Nature Reviews. Disease Primers. 3 (1): 17013. doi:10.1038/nrdp.2017.13. PMID 28332488. S2CID 11605091.
- ^ Heinzel S, Berg D, Gasser T, Chen H, Yao C, Postuma RB (October 2019). "Update of the MDS research criteria for prodromal Parkinson's disease". Movement Disorders. 34 (10): 1464–1470. doi:10.1002/mds.27802. PMID 31412427. S2CID 199663713.
- ^ Sudhakar V, Richardson RM (January 2019). "Gene Therapy for Neurodegenerative Diseases". Neurotherapeutics. 16 (1): 166–175. doi:10.1007/s13311-018-00694-0. PMC 6361055. PMID 30542906.
- ^ Hitti FL, Yang AI, Gonzalez-Alegre P, Baltuch GH (September 2019). "Human gene therapy approaches for the treatment of Parkinson's disease: An overview of current and completed clinical trials". Parkinsonism & Related Disorders. 66: 16–24. doi:10.1016/j.parkreldis.2019.07.018. PMID 31324556. S2CID 198132349.
- ^ Volc D, Poewe W, Kutzelnigg A, et al. (July 2020). "Safety and immunogenicity of the α-synuclein active immunotherapeutic PD01A in patients with Parkinson's disease: a randomised, single-blinded, phase 1 trial". The Lancet. Neurology. 19 (7): 591–600. doi:10.1016/S1474-4422(20)30136-8. PMID 32562684. S2CID 219947651.
- ^ "World's first Parkinson's vaccine is trialled". New Scientist. London. 7 June 2012. Archived from the original on 23 April 2015.
- ^ Jankovic J, Goodman I, Safirstein B, et al. (October 2018). "Safety and Tolerability of Multiple Ascending Doses of PRX002/RG7935, an Anti-α-Synuclein Monoclonal Antibody, in Patients With Parkinson Disease: A Randomized Clinical Trial". JAMA Neurology. 75 (10): 1206–1214. doi:10.1001/jamaneurol.2018.1487. PMC 6233845. PMID 29913017.
- ^ Parmar 2020, pp. 103.
- ^ Parmar 2020, pp. 103–104.
- ^ Parmar 2020, pp. 106.
- ^ Henchcliffe 2018, pp. 134.
- ^ Parmar 2020, pp. 106, 108.
- ^ Schweitzer 2020, pp. 1926.
- ^ Parmar 2020, pp. 105, 109.
- ^ Henchcliffe 2018, pp. 132.
- ^ Jenner P (2014). "An overview of adenosine A2A receptor antagonists in Parkinson's disease". International Review of Neurobiology. 119: 71–86. doi:10.1016/B978-0-12-801022-8.00003-9. ISBN 978-0-12-801022-8. ISSN 2162-5514. PMID 25175961.
- ^ a b Office of the Commissioner (20 February 2020). "FDA approves new add-on drug to treat off episodes in adults with Parkinson's disease". FDA. Retrieved 23 February 2020.
- ^ Blauwendraat 2019, pp. 2.
Works cited edit
Books edit
- Cooper G, Eichhorn G, Rodnitzky RL (2008). "Parkinson's disease". In Conn PM (ed.). Neuroscience in medicine. Humana Press. ISBN 978-1-60327-454-8.
- Dissanayaka NN (8 March 2021). "Chapter 9: Anxiety in Parkinson's Disease". In Byrne GJ, Panchana NA (eds.). Anxiety in Older People: Clinical and Research Perspectives. Cambridge University Press. pp. 139–156. doi:10.1017/9781139087469.009. ISBN 978-1-108-82636-5. S2CID 87250745.
- Ferri FF (2010). "Chapter P". Ferri's differential diagnosis: a practical guide to the differential diagnosis of symptoms, signs, and clinical disorders (2nd ed.). Elsevier/Mosby. ISBN 978-0-323-07699-9.
- O'Sullivan SB, Schmitz TJ (2007). "Parkinson's Disease". Physical Rehabilitation (5th ed.). F.A. Davis. ISBN 978-0-8036-1247-1.
- Simon RP, Greenberg D, Aminoff MJ (2017). Lange Clinical Neurology (10th ed.). McGraw-Hill. ISBN 978-1-259-86172-7.
- Stoker TB, Greenland JC, eds. (December 2018). Parkinson's Disease: Pathogenesis and Clinical Aspects. Codon Publications. ISBN 978-0-9944381-6-4.
- Dallapiazza RF, De Vloo PD, Fomenko A, Lee DJ, Hamani C, Munhoz RP, Hodaie M, Lozano AM, Fasano A, Kalia SK (2018). "Chapter 8: Considerations for Patient and Target Selection in Deep Brain Stimulation surgery for Parkinson's disease". In Stoker TB, Greenland JC (eds.). Parkinson's disease: Pathogenesis and Clinical Aspects. Codon Publications. doi:10.15586/codonpublications.parkinsonsdisease.2018.ch8. ISBN 978-0-9944381-6-4. PMID 30702838. S2CID 81155324.
- Greenland JC, Barker RA (2018). "Chapter 6: The Differential Diagnosis of Parkinson's Disease". In Stoker TB, Greenland JC (eds.). Parkinson's disease: Pathogenesis and Clinical Aspects. Codon Publications. pp. 109–128. doi:10.15586/codonpublications.parkinsonsdisease.2018.ch6. ISBN 978-0-9944381-6-4. PMID 30702839. S2CID 80908095.
- Stoker TB, Torsney KM, Barker RA (2018). "Chapter 3: Pathological mechanisms and clinical aspects of GBA1 mutation-associated Parkinson's disease". In Stoker TB, Greenland JC (eds.). Parkinson's Disease: Pathogenesis and clinical aspects. pp. 45–64. doi:10.15586/codonpublications.parkinsonsdisease.2018.ch3. ISBN 978-0-9944381-6-4. PMID 30702840. S2CID 92170834.
- Tolosa E, Jankovic E, eds. (2007). Parkinson's disease and movement disorders. Lippincott Williams & Wilkins. ISBN 978-0-7817-7881-7.
- Dickson DV (2007). "Neuropathology of movement disorders". In Tolosa E, Jankovic JJ (eds.). Parkinson's disease and movement disorders. Lippincott Williams & Wilkins. ISBN 978-0-7817-7881-7.
- Fung VS, Thompson PD (2007). "Rigidity and spasticity". In Tolosa E, Jankovic E (eds.). Parkinson's disease and movement disorders. Lippincott Williams & Wilkins. ISBN 978-0-7817-7881-7.
- Tolosa E, Katzenschlager R (2007). "Pharmacological management of Parkinson's disease". In Tolosa E, Jankovic JJ (eds.). Parkinson's disease and movement disorders. Lippincott Williams & Wilkins. ISBN 978-0-7817-7881-7.
- Truong DD, Bhidayasiri R (2016). "50: Parkinson's disease". In Lisak RP, Truong DD, Carroll WM, Bhidayasiri R (eds.). International Neurology. John Wiley & Sons. ISBN 978-1-118-77736-7.
Journal articles edit
- Binde CD, Tvete IF, Gåsemyr J, Natvig B, Klemp M (21 May 2018). "A multiple treatment comparison meta-analysis of monoamine oxidase type B inhibitors for Parkinson's disease". British Journal of Clinical Pharmacology. 84 (9): 1917–1927. doi:10.1111/bcp.13651. PMC 6089809. PMID 29847694.
- De Miranda BR, Goldman SM, Miller GW, Greenamy T, Dorseye ER (2022). "Preventing Parkinson's Disease: An Environmental Agenda". Journal of Parkinson's Disease. 12 (1): 45–68. doi:10.3233/JPD-212922. PMC 8842749. PMID 34719434. S2CID 240235393.
- Henchcliffe C, Parmar M (2018). "Repairing the Brain: Cell Replacement Using Stem Cell-Based Technologies". Journal of Parkinson's Disease. 8 (s1): S131–S137. doi:10.3233/JPD-181488. PMC 6311366. PMID 30584166.
- Panicker N, Ge P, Dawson V, Dawson T (April 2021). "The cell biology of Parkinson's disease". Journal of Cell Biology. 220 (4). doi:10.1083/jcb.202012095. PMC 8103423. PMID 33749710.
- Parmar M, Shane G, Claire H (February 2020). "The future of stem cell therapies for Parkinson disease". Nature Reviews Neuroscience. 21 (2): 103–115. doi:10.1038/s41583-019-0257-7. PMID 31907406.
- Polissidis A, Petropoulou-Vathi L, Nakos-Bimpos M, Rideout H (June 2020). "The Future of Targeted Gene-Based Treatment Strategies and Biomarkers in Parkinson's Disease". Biomolecules. 10 (6): 912. doi:10.3390/biom10060912. PMC 7355671. PMID 32560161.
- Schweitzer JS, Song B, Herrington TM, et al. (May 2020). "Personalized iPSC-Derived Dopamine Progenitor Cells for Parkinson's Disease". The New England Journal of Medicine. 382 (20): 1926–1932. doi:10.1056/NEJMoa1915872. PMC 7288982. PMID 32402162.
- Tolosa E, Garrido A, Scholz S, Poewe W (May 2021). "Challenges in the diagnosis of Parkinson's disease". Lancet Neurology. 20 (5): 385–397. doi:10.1016/S1474-4422(21)00030-2. PMC 8185633. PMID 33894193.
- Blauwendraat C, Nalls M, Singleton A (February 2020). "The genetic architecture of Parkinson's disease". Lancet Neurology. 19 (2): 170–178. doi:10.1016/S1474-4422(19)30287-X. PMC 8972299. PMID 31521533.
- Winiker K, Kertscher B (2023). "Behavioural interventions for swallowing in subjects with Parkinson's disease: A mixed methods systematic review". Int J Lang Commun Disord. 58 (4): 1375–1404. doi:10.1111/1460-6984.12865. PMID 36951546. This article incorporates text from this source, which is available under the CC BY 4.0 license.
- Ascherio A, Schwarzschild MA (November 2016). "The epidemiology of Parkinson's disease: risk factors and prevention". Lancet Neurology. 15 (12): 1257–1272. doi:10.1016/S1474-4422(16)30230-7. PMID 27751556.
- De Miranda BR, Goldman SM, Miller GW, Greenamyre JT, Dorsey ER (2022). "Preventing Parkinson's Disease: An Environmental Agenda". Journal of Parkinson's Disease. 12 (1): 45–68. doi:10.3233/JPD-212922. PMC 8842749. PMID 34719434.
- Islam MS, Azim F, Saju H, Zargaran A, Shirzad M, Kamal M, Fatema K, Rehman S, Azad MA, Ebrahimi-Barough S (2021). "Pesticides and Parkinson's disease: Current and future perspective". Journal of Chemical Neuroanatomy. 115. doi:10.1016/j.jchemneu.2021.101966. PMC 8842749. PMID 34719434.
External links edit
- "The Lancet Neurology Video". thelancet.com. Retrieved 27 December 2023. Neurology-related videos, including several showing people with Parkinson's disease.