
Summary
In organic chemistry, a phosphite ester or organophosphite usually refers to an organophosphorous compound with the formula P(OR)3. They can be considered as esters of an unobserved tautomer phosphorous acid, H3PO3, with the simplest example being trimethylphosphite, P(OCH3)3. Some phosphites can be considered esters of the dominant tautomer of phosphorous acid (HP(O)(OH)2). The simplest representative is dimethylphosphite with the formula HP(O)(OCH3)2. Both classes of phosphites are usually colorless liquids.
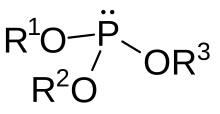
Synthesis edit
- From PCl3
Phosphite esters are typically prepared by treating phosphorus trichloride with an alcohol. For alkyl alcohols the displaced chloride ion can attack the phosphite, causing dealkylation to give a dialkylphosphite and an organochlorine compound.[1][2] The overall reaction is as follows:
- PCl3 + 3 C2H5OH → (C2H5O)2P(O)H + 2 HCl + C2H5Cl
Alternatively, when the alcoholysis is conducted in the presence of proton acceptors (typically an amine base), one obtains the C3-symmetric trialkyl derivatives:[3]
- PCl3 + 3 C2H5OH + 3 R3N → (C2H5O)3P + 3 R3NHCl
A base is not essential when using aromatic alcohols such as phenols, as they are not susceptible to attack by chloride, however it does catalyse the esterification reaction and is therefore often included.[4]
- By transesterification
Phosphite esters can also be prepared by transesterification, as they undergo alcohol exchange upon heating with other alcohols.[5] This process is reversible and can be used to produce mixed alkyl phosphites. Alternatively, if the phosphite of a volatile alcohol is used, such as trimethyl phosphite, then the by product (methanol) can be removed by distillation, allowing the reaction to be driven to completion.
Reactions and applications of tris(organo)phosphites edit
Reactions edit
Tris(2,4-di-tert-butylphenyl)phosphite, a widely used stabilizer in polymers
Phosphites are oxidized to phosphate esters:
- P(OR)3 + [O] → OP(OR)3
This reaction underpins the commercial use of some phosphite esters as stabilizers in polymers.[6]
Alkyl phosphite esters are used in the Perkow reaction for the formation of vinyl phosphonates, and in the Michaelis–Arbuzov reaction to form phosphonates. Aryl phosphite esters may not undergo these reactions and hence are commonly used as stabilizers in halogen-bearing polymers such as PVC.
Phosphite esters may be used as reducing agents in more specialised cases. For example, triethylphosphite is known to reduce certain hydroperoxides to alcohols formed by autoxidation[7] (scheme). In this process the phosphite is converted to a phosphate ester. This reaction type is also utilized in the Wender Taxol total synthesis.
Homogeneous catalysis edit
Phosphite esters are Lewis bases and hence can form coordination complexes with various metal ions. Representative phosphite ligands include trimethylphosphite ((MeO)3P), triethylphosphite ((EtO)3P), trimethylolpropane phosphite, and triphenylphosphite ((PhO)3P). Phosphites exhibit a smaller ligand cone angles than the structurally related phosphine ligand family. Phosphite ligands are components of industrial catalysts for hydroformylation and hydrocyanation.[8]
Chemistry of HP(O)(OR)2 edit
Structure of a diethylphosphite.
Diorganophosphites are derivatives of phosphorus(V) and can be viewed as the di-esters of phosphorous acid ((HO)2P(O)H). They exhibit tautomerism, however, the equilibrium overwhelmingly favours the right-hand (phosphonate-like) form:[11][12]
- (RO)2POH ⇌ (RO)2P(O)H
The P-H bond is the site of high reactivity in these compounds (for example in the Atherton–Todd reaction and Hirao coupling), whereas in tri-organophosphites the lone pair on phosphorus is the site of high reactivity. Diorganophosphites do however undergo transesterification.
See also edit
- Phosphinite P(OR)R2
- Phosphonite P(OR)2R
- Ortho ester CH(OR)3
- Borate ester B(OR)3
References edit
- ^ Malowan, John E. (1953). "Diethyl phosphite". Inorganic Syntheses. Inorganic Syntheses. Vol. 4. pp. 58–60. doi:10.1002/9780470132357.ch19. ISBN 9780470132357.
- ^ Gerrard, W.; Whitbread, E. G. G. (1952). "165. Interaction of hydrogen halides and n-butyl phosphites". Journal of the Chemical Society (Resumed): 914. doi:10.1039/JR9520000914.
- ^ A. H. Ford-Moore & B. J. Perry (1963). "Triethyl Phosphite". Organic Syntheses; Collected Volumes, vol. 4, p. 955.
- ^ Mao, Mengmei; Zhang, Le; Yao, Hanlin; Wan, Li; Xin, Zhong (28 April 2020). "Development and Scale-up of the Rapid Synthesis of Triphenyl Phosphites in Continuous Flow". ACS Omega. 5 (16): 9503–9509. doi:10.1021/acsomega.0c00716. PMC 7191834. PMID 32363302.
- ^ Hoffmann, Friedrich W.; Ess, Richard J.; Usingef, Robert P. (November 1956). "The Transesterification of Trialkyl Phosphites with Aliphatic Alcohols". Journal of the American Chemical Society. 78 (22): 5817–5821. doi:10.1021/ja01603a026.
- ^ Rainer Wolf; Bansi Lal Kaul (2000). "Plastics, Additives". Ullmann's Encyclopedia Of Industrial Chemistry. doi:10.1002/14356007.a20_459. ISBN 3527306730.
- ^ J. N. Gardner; F. E. Carlon & O. Gnoj (1968). "One-step procedure for the preparation of tertiary α-ketols from the corresponding ketones". J. Org. Chem. 33 (8): 3294–3297. doi:10.1021/jo01272a055. PMID 5742870.
- ^ Aitor Gual; Cyril Godard; Verónica de la Fuente; Sergio Castillón (2012). "Design and Synthesis of Phosphite Ligands for Homogeneous Catalysis". In Paul C. J. Kamer; Piet W. N. M. van Leeuwen (eds.). Phosphorus(III) Ligands in Homogeneous Catalysis: Design and Synthesis. John Wiley & Sons. pp. 81–131. doi:10.1002/9781118299715.ch3. ISBN 9781118299715.
- ^ Cuny, Gregory D.; Buchwald, Stephen L. (1993). "Practical, High-Yield, Regioselective, Rhodium-Catalyzed Hydroformylation of Functionalized α-olefins". Journal of the American Chemical Society. 115 (5): 2066–2068. doi:10.1021/ja00058a079.
- ^ Van Rooy, Annemiek; Kamer, Paul C. J.; Van Leeuwen, Piet W. N. M.; Goubitz, Kees; Fraanje, Jan; Veldman, Nora; Spek, Anthony L. (1996). "Bulky Diphosphite-Modified Rhodium Catalysts: Hydroformylation and Characterization". Organometallics. 15 (2): 835–847. doi:10.1021/OM950549K.
- ^ Guthrie, J. Peter (1979). "Tautomerization Equilibria for Phosphorous Acid and its Ethyl Esters, Free Energies of Formation of Phosphorous and Phosphonic Acids and their Ethyl Esters, and p Ka Values for Ionization of the P—H Bond in Phosphonic Acid and Phosphonic Esters". Canadian Journal of Chemistry. 57 (2): 236–239. doi:10.1139/v79-039.
- ^ Janesko, Benjamin G.; Fisher, Henry C.; Bridle, Mark J.; Montchamp, Jean-Luc (2015-09-29). "P(═O)H to P–OH Tautomerism: A Theoretical and Experimental Study". The Journal of Organic Chemistry. 80 (20). American Chemical Society (ACS): 10025–10032. doi:10.1021/acs.joc.5b01618. ISSN 0022-3263. PMID 26372089.


