
Summary
Protocadherins (Pcdhs) are the largest mammalian subgroup of the cadherin superfamily of homophilic cell-adhesion proteins.[1] They were discovered by Shintaro Suzuki's group, when they used PCR to find new members of the cadherin family. The PCR fragments that corresponded to protocadherins were found in vertebrate and invertebrate species.[2] This prevalence in a wide range of species suggested that the fragments were part of an ancient cadherin and were thus termed "Protocadherins" as the "first cadherins". Of the approximately 70 Pcdh genes identified in mammalian genomes, over 50 are located in tightly linked gene clusters on the same chromosome.[3] Until recently, it was assumed that this kind of organization can only be found in vertebrates,[3] but Octopus bimaculoides has 168 genes of which nearly three-quarters are found in tandem clusters with the two largest clusters compromising 31 and 17 genes, respectively.[4]
| Protocadherin, cytoplasmic | |||||||||
|---|---|---|---|---|---|---|---|---|---|
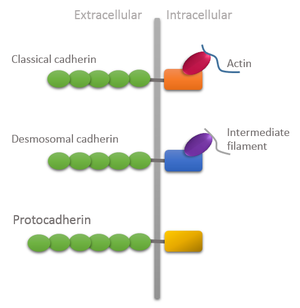 Domain organization of different types of cadherins showing unique features of protocadherins: Extracellular domain is longer and intracellular domain lacks attachment with cytoskeleton. | |||||||||
| Identifiers | |||||||||
| Symbol | PDCH | ||||||||
| Pfam | PF08374 | ||||||||
| InterPro | IPR013585 | ||||||||
| Membranome | 114 | ||||||||
| |||||||||
Classification edit
In mammals, two types of Pcdh genes have been defined: the non-clustered Pcdhs which are scattered throughout the genome; and the clustered Pcdhs organized in three gene clusters designated α, β, γ which in mouse genome comprises 14, 22 and 22, respectively, large variable exons arrayed in tandem. Each exon is transcribed from its owner promoter and encodes: the entire extracellular domain, a transmembrane domain, and a short and variable intracellular domain of the corresponding Pcdh protein which differs from the Cadherin intracellular domain due to lack of attachment to the cytoskeleton through catenins.[5]
Moreover, these clustered Pcdh genes are predominantly expressed in the developing nervous system[2] and since different subsets of Pcdhs genes are differentially expressed in individual neurons, a vast cell surface diversity may arise from this combinatorial expression.[5] This has led to speculation and further to the proposal that Pcdhs may provide a synaptic-address code for neuronal connectivity or a single-cell barcode for self-recognition/self-avoidance similar to that ascribed to DSCAM proteins of invertebrates. Although vertebrate DSCAMs lack the diversity of their invertebrate counterparts, the selective transcription of individual Pcdh isoforms can be achieved by promoter choice followed by alternative pre-mRNA cis-splicing thus increasing the number of possible combinations.
Function edit
Homophilic interactions and intracellular signaling edit
Clustered Pcdhs proteins are detected throughout the neuronal soma, dendrites and axons and are observed in synapses and growth cones.[6][7][8][9][10] Like classical cadherins, members of Pcdhs family were also shown to mediate cell-cell adhesion in cell-based assays[11][12][13] and most of them showed to engage in homophilic trans-interactions.[14] Schreiner and Weiner [14] showed that Pcdhα and γ proteins can form multimeric complexes. If all three classes of Pcdhs could engage in multimerization of stochastically expressed Pcdhs isoforms, then neurons could produce a large number of distinct homophilic interaction units, amplifying significantly the cell-surface diversity more than the one afforded by stochastic gene expression alone.
As for cytoplasmic domain, all the three classes of clustered Pcdhs proteins are dissimilar, although they are strictly conserved in vertebrate evolution, suggesting a conserved cellular function.[5] This is corroborated by a large number of other interacting proteins including phosphatases, kinases, adhesion molecules and synaptic proteins[15] The cytoplasmic domain also mediates intracellular retention, a property which distinguishes the clustered protocadherins from the related classical cadherins.[16] Furthermore, it was shown that Pcdhs are proteolytically processed by γ-secretase complex,[17][18] which releases soluble intracellular fragments into the cytoplasm which might have a broad range of functions as acting locally in the cytoplasm and/or even regulate gene expression similarly to other cell-surface proteins such as Notch and N-cadherin. Since these molecules are involved in so many developmental processes like axon guidance and dendrite arborization, mutations in Pcdhs genes and their expression may play a role in Down, Rett as well as Fragile X syndrome,[19] schizophrenia,[20] and neurodegenerative diseases[21]
The cytoplasmic domain of Pcdh-alpha can be divided into two specific types. Both of them enhance homophilic interactions. They associate with neurofillament M and fascin respectively.[22]
Human genes edit
- PCDH1
- PCDH7
- PCDH8
- PCDH9
- PCDH10
- PCDH11X/11Y
- PCDH12
- PCDH15
- PCDH17
- PCDH18
- PCDH19
- PCDH20
- PCDHA1
- PCDHA2
- PCDHA3
- PCDHA4
- PCDHA5
- PCDHA6
- PCDHA7
- PCDHA8
- PCDHA9
- PCDHA10
- PCDHA11
- PCDHA12
- PCDHA13
- PCDHAC1
- PCDHAC2
- PCDHB1
- PCDHB2
- PCDHB3
- PCDHB4
- PCDHB5
- PCDHB6
- PCDHB7
- PCDHB8
- PCDHB9
- PCDHB10
- PCDHB11
- PCDHB12
- PCDHB13
- PCDHB14
- PCDHB15
- PCDHB16
- PCDHB17
- PCDHB18
- PCDHGA1
- PCDHGA2
- PCDHGA3
- PCDHGA4
- PCDHGA5
- PCDHGA6
- PCDHGA7
- PCDHGA8
- PCDHGA9
- PCDHGA10
- PCDHGA11
- PCDHGA12
- PCDHGB1
- PCDHGB2
- PCDHGB3
- PCDHGB4
- PCDHGB5
- PCDHGB6
- PCDHGB7
- PCDHGC3
- PCDHGC4
- PCDHGC5
- FAT
- FAT2
- FAT4
See also edit
- Cadherin
- PCDH11X
- Neuronal self-avoidance
- Epileptic Encephalopathy, Early Infantile, 9, caused by mutation in the gene encoding protocadherin-19
References edit
- ^ Hulpiau P, van Roy F (February 2009). "Molecular evolution of the cadherin superfamily". The International Journal of Biochemistry & Cell Biology. 41 (2): 349–69. doi:10.1016/j.biocel.2008.09.027. PMID 18848899.
- ^ a b Sano K, Tanihara H, Heimark RL, Obata S, Davidson M, St John T, Taketani S, Suzuki S (June 1993). "Protocadherins: a large family of cadherin-related molecules in central nervous system". The EMBO Journal. 12 (6): 2249–56. doi:10.1002/j.1460-2075.1993.tb05878.x. PMC 413453. PMID 8508762.
- ^ a b Chen WV, Alvarez FJ, Lefebvre JL, Friedman B, Nwakeze C, Geiman E, Smith C, Thu CA, Tapia JC, Tasic B, Sanes JR, Maniatis T (August 2012). "Functional significance of isoform diversification in the protocadherin gamma gene cluster". Neuron. 75 (3): 402–9. doi:10.1016/j.neuron.2012.06.039. PMC 3426296. PMID 22884324.
- ^ Albertin, Caroline B.; Simakov, Oleg; Mitros, Therese; Wang, Z. Yan; Pungor, Judit R.; Edsinger-Gonzales, Eric; Brenner, Sydney; Ragsdale, Clifton W.; Rokhsar, Daniel S. (August 2015). "The octopus genome and the evolution of cephalopod neural and morphological novelties". Nature. 524 (7564): 220–224. Bibcode:2015Natur.524..220A. doi:10.1038/nature14668. ISSN 0028-0836. PMC 4795812. PMID 26268193.
- ^ a b c Chen WV, Maniatis T (August 2013). "Clustered protocadherins". Development. 140 (16): 3297–302. doi:10.1242/dev.090621. PMC 3737714. PMID 23900538.
- ^ Kohmura N, Senzaki K, Hamada S, Kai N, Yasuda R, Watanabe M, Ishii H, Yasuda M, Mishina M, Yagi T (June 1998). "Diversity revealed by a novel family of cadherins expressed in neurons at a synaptic complex". Neuron. 20 (6): 1137–51. doi:10.1016/s0896-6273(00)80495-x. PMID 9655502.
- ^ Wang X, Weiner JA, Levi S, Craig AM, Bradley A, Sanes JR (December 2002). "Gamma protocadherins are required for survival of spinal interneurons". Neuron. 36 (5): 843–54. doi:10.1016/s0896-6273(02)01090-5. PMID 12467588.
- ^ Kallenbach S, Khantane S, Carroll P, Gayet O, Alonso S, Henderson CE, Dudley K (June 2003). "Changes in subcellular distribution of protocadherin gamma proteins accompany maturation of spinal neurons". Journal of Neuroscience Research. 72 (5): 549–56. doi:10.1002/jnr.10618. PMID 12749019. S2CID 41159576.
- ^ Phillips GR, Tanaka H, Frank M, Elste A, Fidler L, Benson DL, Colman DR (June 2003). "Gamma-protocadherins are targeted to subsets of synapses and intracellular organelles in neurons". The Journal of Neuroscience. 23 (12): 5096–104. doi:10.1523/JNEUROSCI.23-12-05096.2003. PMC 6741188. PMID 12832533.
- ^ Junghans D, Heidenreich M, Hack I, Taylor V, Frotscher M, Kemler R (February 2008). "Postsynaptic and differential localization to neuronal subtypes of protocadherin beta16 in the mammalian central nervous system". The European Journal of Neuroscience. 27 (3): 559–71. doi:10.1111/j.1460-9568.2008.06052.x. PMID 18279309. S2CID 24229102.
- ^ Obata S, Sago H, Mori N, Rochelle JM, Seldin MF, Davidson M, St John T, Taketani S, Suzuki ST (December 1995). "Protocadherin Pcdh2 shows properties similar to, but distinct from, those of classical cadherins". Journal of Cell Science. 108 ( Pt 12) (12): 3765–73. doi:10.1242/jcs.108.12.3765. PMID 8719883.
- ^ Frank M, Ebert M, Shan W, Phillips GR, Arndt K, Colman DR, Kemler R (August 2005). "Differential expression of individual gamma-protocadherins during mouse brain development". Molecular and Cellular Neurosciences. 29 (4): 603–16. doi:10.1016/j.mcn.2005.05.001. PMID 15964765. S2CID 40074917.
- ^ Reiss K, Maretzky T, Haas IG, Schulte M, Ludwig A, Frank M, Saftig P (August 2006). "Regulated ADAM10-dependent ectodomain shedding of gamma-protocadherin C3 modulates cell-cell adhesion". The Journal of Biological Chemistry. 281 (31): 21735–44. doi:10.1074/jbc.M602663200. PMID 16751190.
- ^ a b Schreiner D, Weiner JA (August 2010). "Combinatorial homophilic interaction between gamma-protocadherin multimers greatly expands the molecular diversity of cell adhesion". Proceedings of the National Academy of Sciences of the United States of America. 107 (33): 14893–8. Bibcode:2010PNAS..10714893S. doi:10.1073/pnas.1004526107. PMC 2930437. PMID 20679223.
- ^ Schalm SS, Ballif BA, Buchanan SM, Phillips GR, Maniatis T (August 2010). "Phosphorylation of protocadherin proteins by the receptor tyrosine kinase Ret". Proceedings of the National Academy of Sciences of the United States of America. 107 (31): 13894–9. doi:10.1073/pnas.1007182107. PMC 2922223. PMID 20616001.
- ^ Fernández-Monreal M, Kang S, Phillips GR (March 2009). "Gamma-protocadherin homophilic interaction and intracellular trafficking is controlled by the cytoplasmic domain in neurons". Molecular and Cellular Neurosciences. 40 (3): 344–53. doi:10.1016/j.mcn.2008.12.002. PMC 2646808. PMID 19136062.
- ^ Bonn S, Seeburg PH, Schwarz MK (June 2007). "Combinatorial expression of alpha- and gamma-protocadherins alters their presenilin-dependent processing". Molecular and Cellular Biology. 27 (11): 4121–32. doi:10.1128/MCB.01708-06. PMC 1900011. PMID 17403907.
- ^ Buchanan SM, Schalm SS, Maniatis T (October 2010). "Proteolytic processing of protocadherin proteins requires endocytosis". Proceedings of the National Academy of Sciences of the United States of America. 107 (41): 17774–9. Bibcode:2010PNAS..10717774B. doi:10.1073/pnas.1013105107. PMC 2955128. PMID 20876099.
- ^ Kaufmann WE, Moser HW (October 2000). "Dendritic anomalies in disorders associated with mental retardation". Cerebral Cortex. 10 (10): 981–91. doi:10.1093/cercor/10.10.981. PMID 11007549.
- ^ Kalmady SV, Venkatasubramanian G (March 2009). "Evidence for positive selection on Protocadherin Y gene in Homo sapiens: implications for schizophrenia". Schizophrenia Research. 108 (1–3): 299–300. doi:10.1016/j.schres.2008.09.015. PMID 18938061. S2CID 26590444.
- ^ Anderton BH, Callahan L, Coleman P, Davies P, Flood D, Jicha GA, Ohm T, Weaver C (August 1998). "Dendritic changes in Alzheimer's disease and factors that may underlie these changes". Progress in Neurobiology. 55 (6): 595–609. doi:10.1016/s0301-0082(98)00022-7. PMID 9670220. S2CID 11421820.
- ^ Triana-Baltzer GB, Blank M (March 2006). "Cytoplasmic domain of protocadherin-alpha enhances homophilic interactions and recognizes cytoskeletal elements". Journal of Neurobiology. 66 (4): 393–407. doi:10.1002/neu.20228. PMID 16408303.
Further reading edit
- Han MH, Lin C, Meng S, Wang X (January 2010). "Proteomics analysis reveals overlapping functions of clustered protocadherins". Molecular & Cellular Proteomics. 9 (1): 71–83. doi:10.1074/mcp.M900343-MCP200. PMC 2808268. PMID 19843561.
- Sotomayor M, Gaudet R, Corey DP (September 2014). "Sorting out a promiscuous superfamily: towards cadherin connectomics". Trends in Cell Biology. 24 (9): 524–36. doi:10.1016/j.tcb.2014.03.007. PMC 4294768. PMID 24794279.
- Pancho A, Aerts T, Mitsogiannis MD, Seuntjens E (June 2020). "PProtocadherins at the Crossroad of Signaling Pathways". Frontiers in Molecular Neuroscience. 13: 117. doi:10.3389/fnmol.2020.00117. PMC 7339444. PMID 32694982.


