
KNOWPIA
WELCOME TO KNOWPIA
Summary
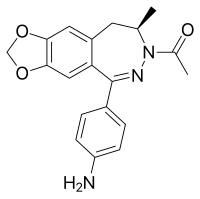
Talampanel (INN; development codes GYKI 537773 and LY300164) is a drug which has been investigated for the treatment of epilepsy,[1][2] malignant gliomas,[3] and amyotrophic lateral sclerosis (ALS).[4]
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID |
|
| ChemSpider |
|
| UNII |
|
| ChEMBL |
|
| CompTox Dashboard (EPA) |
|
| ECHA InfoCard | 100.230.001 |
| Chemical and physical data | |
| Formula | C19H19N3O3 |
| Molar mass | 337.379 g·mol−1 |
| 3D model (JSmol) |
|
| |
| |
| | |
As of May 2010, results from the trial for ALS have been found negative.[5] Talampanel is not currently under development.
Talampanel acts as a non-competitive antagonist of the AMPA receptor, a type of ionotropic glutamate receptor in the central nervous system.[6]
It showed effectiveness for epilepsy in clinical trials but its development was suspended due to its poor pharmacokinetic profile, namely a short terminal half-life (3 hours) that necessitated multiple doses per day.[7]
References edit
- ^ Luszczki JJ (2009). "Third-generation antiepileptic drugs: mechanisms of action, pharmacokinetics and interactions". Pharmacological Reports. 61 (2): 197–216. doi:10.1016/s1734-1140(09)70024-6. PMID 19443931. S2CID 72918370.
- ^ Bialer M, Johannessen SI, Kupferberg HJ, Levy RH, Perucca E, Tomson T (January 2007). "Progress report on new antiepileptic drugs: a summary of the Eigth [sic] Eilat Conference (EILAT VIII)". Epilepsy Research. 73 (1): 1–52. doi:10.1016/j.eplepsyres.2006.10.008. PMID 17158031. S2CID 45026113.
- ^ Iwamoto FM, Kreisl TN, Kim L, Duic JP, Butman JA, Albert PS, Fine HA (April 2010). "Phase 2 trial of talampanel, a glutamate receptor inhibitor, for adults with recurrent malignant gliomas". Cancer. 116 (7): 1776–1782. doi:10.1002/cncr.24957. PMC 2846997. PMID 20143438.
- ^ Pascuzzi RM, Shefner J, Chappell AS, Bjerke JS, Tamura R, Chaudhry V, et al. (May 2010). "A phase II trial of talampanel in subjects with amyotrophic lateral sclerosis". Amyotrophic Lateral Sclerosis. 11 (3): 266–271. doi:10.3109/17482960903307805. PMID 19961264. S2CID 7388452.
- ^ "Talampanel Trial". alsa.org. May 2010. Archived from the original on 2011-03-23.
- ^ Aujla PK, Fetell MR, Jensen FE (April 2009). "Talampanel suppresses the acute and chronic effects of seizures in a rodent neonatal seizure model". Epilepsia. 50 (4): 694–701. doi:10.1111/j.1528-1167.2008.01947.x. PMC 2672962. PMID 19220413.
- ^ Lee K, Goodman L, Fourie C, Schenk S, Leitch B, Montgomery JM (2016). "AMPA Receptors as Therapeutic Targets for Neurological Disorders". Ion Channels as Therapeutic Targets, Part A. Advances in Protein Chemistry and Structural Biology. Vol. 103. pp. 203–261. doi:10.1016/bs.apcsb.2015.10.004. ISBN 9780128047941. PMID 26920691.


