
Summary
Tris(dibenzylideneacetone)dipalladium(0) or [Pd2(dba)3] is an organopalladium compound. The compound is a complex of palladium(0) with dibenzylideneacetone (dba). It is a dark-purple/brown solid, which is modestly soluble in organic solvents. Because the dba ligands are easily displaced, the complex is used as a homogeneous catalyst in organic synthesis.[1]
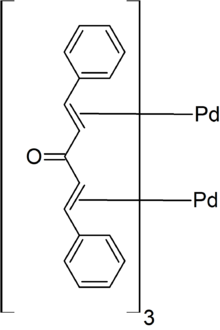
| |

| |
| Names | |
|---|---|
| IUPAC name
Tris(dibenzylideneacetone)dipalladium
| |
| Other names
Pd2(dba)3
| |
| Identifiers | |
| |
| ECHA InfoCard | 100.122.794 |
PubChem CID
|
|
| Properties | |
| C51H42O3Pd2 | |
| Molar mass | 915.73 g·mol−1 |
| Melting point | 152 to 155 °C (306 to 311 °F; 425 to 428 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Preparation and structure edit
First reported in 1970,[2] it is prepared from dibenzylideneacetone and sodium tetrachloropalladate. Because it is commonly recrystallized from chloroform, the complex is often supplied as the adduct [Pd2(dba)3·CHCl3].[1] The purity of samples can be variable.[3]
In [Pd2(dba)3], the pair of Pd atoms are separated by 320 pm but are tied together by dba units.[4] The Pd(0) centres are bound to the alkene parts of the dba ligands.
Applications edit
[Pd2(dba)3] is used as a source of soluble Pd(0), in particular as a catalyst for various coupling reactions. Examples of reactions using this reagent are the Negishi coupling, Suzuki coupling, Carroll rearrangement, and Trost asymmetric allylic alkylation, as well as Buchwald–Hartwig amination.[5]
Related Pd(0) complexes are [Pd(dba)2][6] and tetrakis(triphenylphosphine)palladium(0).
References edit
- ^ a b Jiro Tsuji and Ian J. S. Fairlamb "Tris(dibenzylideneacetone)dipalladium–Chloroform" E-EROS, 2008. doi:10.1002/047084289X.rt400.pub2
- ^ Takahashi, Y.; Ito, Ts.; Sakai, S.; Ishii, Y. (1970). "A novel palladium(0) complex; bis(dibenzylideneacetone)palladium(0)". Journal of the Chemical Society D: Chemical Communications (17): 1065. doi:10.1039/C29700001065.
- ^ Zalesskiy, S. S., Ananikov, V. P., "Pd2(dba)3 as a Precursor of Soluble Metal Complexes and Nanoparticles: Determination of Palladium Active Species for Catalysis and Synthesis", Organometallics 2012, 31, 2302. doi:10.1021/om201217r
- ^ Pierpont, Cortlandt G.; Mazza, Margaret C. (1974). "Crystal and molecular structure of tris(dibenzylideneacetone)dipalladium(0)". Inorg. Chem. 13 (8): 1891. doi:10.1021/ic50138a020.
- ^ Hartwig, J. F. (2010). Organotransition Metal Chemistry, from Bonding to Catalysis. New York: University Science Books. ISBN 978-1-891389-53-5.
- ^ John R. Stille, F. Christopher Pigge, Christopher S. Regens, Ke Chen, Adrian Ortiz and Martin D. Eastgate "Bis(dibenzylideneacetone)palladium(0)" E-eros. 2013. doi:10.1002/047084289X.rb138.pub3


