KNOWPIA
WELCOME TO KNOWPIA
Summary
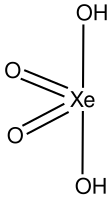
Xenic acid is a proposed noble gas compound with the chemical formula H2XeO4 or XeO2(OH)2. It has not been isolated, and the published characterization data are ambiguous.[1]
| |||
| Identifiers | |||
|---|---|---|---|
3D model (JSmol)
|
| ||
| ChemSpider |
| ||
PubChem CID
|
| ||
| |||
| |||
| Properties | |||
| H2XeO4 | |||
| Molar mass | 197.31 g/mol | ||
| Related compounds | |||
Related compounds
|
Perxenic acid Xenon trioxide | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |||
Salts of xenic acid are called xenates, containing the HXeO−
4 anion, such as monosodium xenate. They tend to disproportionate into xenon gas and perxenates:[2]
- 2 HXeO−
4 + 2 OH−
→ XeO4−
6 + Xe + O
2 + 2 H
2O
The energy given off is sufficient to form ozone from diatomic oxygen:
- 3 O
2 (g) → 2 O
3 (g)
Salts containing the deprotonated anion XeO2−
4 are presently unknown.[2]
References edit
Further reading edit
- Bruno Jaselskis, Stanislaus Vas (May 1964). "Xenic Acid Reactions with vic-Diols". J. Am. Chem. Soc. 86 (10): 2078–2079. doi:10.1021/ja01064a041.