
KNOWPIA
WELCOME TO KNOWPIA
Summary
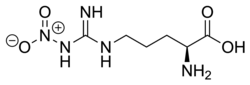
Nitroarginine, or Nω-nitro-l-arginine, also known as L-NOARG, is a nitro derivative of the amino acid arginine.[1] It is an inhibitor of nitric oxide synthase and hence a vasoconstrictor. As such, it finds widespread use as a biochemical tool in the study of nitric oxide and its biological effects.[2]
| |
| Names | |
|---|---|
| IUPAC name
(2S)-2-Amino-5-[[amino(nitramido)methylidene]amino]pentanoic acid
| |
| Other names
N5-(nitroamidino)-l-Ornithine; (+)-NG-Nitroarginine; NG-nitro-l-Arginine, l-NG-Nitroarginine; L-NNA; L-NOARG; NG-Nitro-l-arginine; NG-Nitroarginine; NOLA; NSC 53662; Nitro-l-arginine; Nω-Nitro-l-arginine; Nω-Nitro-l-arginine; ω-Nitro-l-arginine; ω-Nitroarginine
| |
| Identifiers | |
| |
3D model (JSmol)
|
|
| ChEMBL |
|
| ECHA InfoCard | 100.016.745 |
PubChem CID
|
|
| UNII |
|
CompTox Dashboard (EPA)
|
|
| |
| Properties | |
| C6H13N5O4 | |
| Molar mass | 219.201 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Nitroarginine has been used in research studying coronary constriction, and it was found that, in the presence of midazolam vasodilatation was unaffected by nitroarginine.[3] Due to the presence of all three isoforms of nitric oxide synthase in striatal tissue in the forebrain, research has also been conducted on how its inhibition might affect monoamine transport and dopamine half-life in the striatal extracellular space.[4]
References edit
- ^ Moore, P. K.; Al‐Swayeh, O. A.; Chong, N. W. S.; Evans, R. A.; Gibson, A. (1990). "l-NG-nitro arginine (l-NOARG), a novel, l-arginine-reversible inhibitor of endothelium-dependent vasodilatation in vitro". British Journal of Pharmacology. 99 (2): 408–412. doi:10.1111/j.1476-5381.1990.tb14717.x. ISSN 1476-5381. PMC 1917379. PMID 2328404.
- ^ Bansinath M, Arbabha B, Turndorf H, Garg UC (1993). "Chronic administration of a nitric oxide synthase inhibitor, Nω-nitro-L-arginine, and drug-induced increase in cerebellar cyclic GMP in vivo". Neurochemical Research. 18 (10): 1063–6. doi:10.1007/BF00966685. PMID 7504789. S2CID 2447933.
- ^ O.L. Woodman; G.J. Dusting (1991). "N-nitro L-arginine causes coronary vasoconstriction and inhibits endothelium-dependent vasodilatation in anaesthetized greyhounds". Br. J. Pharmacol. 103 (2): 1407–1410. doi:10.1111/j.1476-5381.1991.tb09802.x. PMC 1908370. PMID 1909199.
- ^ Prieto, Sonia Guerrero; Silva, João Carlos dos Santos; de Lima, Mairon Oliveira; Almeida, Maria Camila; Echeverry, Marcela Bermúdez (February 2019). "Cross-tolerance between nitric oxide synthase inhibition and atypical antipsychotics modify nicotinamide-adenine-dinucleotide phosphate-diaphorase activity in mouse lateral striatum". Behavioural Pharmacology. 30 (1): 67–78. doi:10.1097/FBP.0000000000000406. ISSN 0955-8810. PMID 29664745. S2CID 4933721 – via Ovid.


