
Summary
Germane is the chemical compound with the formula GeH4, and the germanium analogue of methane. It is the simplest germanium hydride and one of the most useful compounds of germanium. Like the related compounds silane and methane, germane is tetrahedral. It burns in air to produce GeO2 and water. Germane is a group 14 hydride.
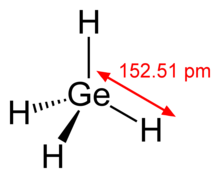
| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Germane
| |||
| Other names
Germanium tetrahydride
Germanomethane Monogermane | |||
| Identifiers | |||
| |||
3D model (JSmol)
|
| ||
| ChEBI |
| ||
| ChemSpider |
| ||
| ECHA InfoCard | 100.029.055 | ||
| EC Number |
| ||
| 587 | |||
| KEGG |
| ||
PubChem CID
|
| ||
| RTECS number |
| ||
| UNII |
| ||
| UN number | 2192 | ||
CompTox Dashboard (EPA)
|
| ||
| |||
| |||
| Properties | |||
| GeH4 | |||
| Molar mass | 76.62 g/mol | ||
| Appearance | Colorless gas | ||
| Odor | Pungent[1] | ||
| Density | 3.3 kg/m3 | ||
| Melting point | −165 °C (−265 °F; 108 K) | ||
| Boiling point | −88 °C (−126 °F; 185 K) | ||
| Low | |||
| Vapor pressure | >1 atm[1] | ||
| Viscosity | 17.21 μPa·s (theoretical estimate)[2] | ||
| Structure | |||
| Tetrahedral | |||
| 0 D | |||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Toxic, flammable, may ignite spontaneously in air | ||
| GHS labelling: | |||
   
| |||
| Danger | |||
| H220, H280, H302, H330 | |||
| P210, P260, P264, P270, P271, P284, P301+P312, P304+P340, P310, P320, P330, P377, P381, P403, P403+P233, P405, P410+P403, P501 | |||
| NFPA 704 (fire diamond) | |||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
None[1] | ||
REL (Recommended)
|
TWA 0.2 ppm (0.6 mg/m3)[1] | ||
IDLH (Immediate danger)
|
N.D.[1] | ||
| Safety data sheet (SDS) | ICSC 1244 | ||
| Related compounds | |||
Related compounds
|
Methane Silane Stannane Plumbane Germyl | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |||
Occurrence edit
Synthesis edit
Germane is typically prepared by reduction of germanium oxides, notably germanates, with hydride reagents such as sodium borohydride, potassium borohydride, lithium borohydride, lithium aluminium hydride, sodium aluminium hydride. The reaction with borohydrides is catalyzed by various acids and can be carried out in either aqueous or organic solvent. On laboratory scale, germane can be prepared by the reaction of Ge(IV) compounds with these hydride reagents.[4][5] A typical synthesis involved the reaction of sodium germanate with potassium borohydride.[6]
- NaHGeO3 + KBH4 + H2O → KGeH3 + KB(OH)4
- KGeH3 + HO2CCH3 → GeH4 + KO2CCH3
Other methods for the synthesis of germane include electrochemical reduction and a plasma-based method.[7] The electrochemical reduction method involves applying voltage to a germanium metal cathode immersed in an aqueous electrolyte solution and an anode counter-electrode composed of a metal such as molybdenum or cadmium. In this method, germane and hydrogen gases evolve from the cathode while the anode reacts to form solid molybdenum oxide or cadmium oxides. The plasma synthesis method involves bombarding germanium metal with hydrogen atoms (H) that are generated using a high frequency plasma source to produce germane and digermane.
Reactions edit
Germane is weakly acidic. In liquid ammonia GeH4 is ionised forming NH4+ and GeH3−.[8] With alkali metals in liquid ammonia GeH4 reacts to give white crystalline MGeH3 compounds. The potassium (potassium germyl or potassium trihydrogen germanide KGeH3) and rubidium compounds (rubidium germyl or rubidium trihydrogen germanide RbGeH3) have the sodium chloride structure implying a free rotation of the trihydrogen germanide anion GeH3−, the caesium compound, caesium germyl or caesium trihydrogen germanide CsGeH3 in contrast has the distorted sodium chloride structure of TlI.[8]
Use in semiconductor industry edit
The gas decomposes near 600K (327°C; 620°F) to germanium and hydrogen. Because of its thermal lability, germane is used in the semiconductor industry for the epitaxial growth of germanium by MOVPE or chemical beam epitaxy.[9] Organogermanium precursors (e.g. isobutylgermane, alkylgermanium trichlorides, and dimethylaminogermanium trichloride) have been examined as less hazardous liquid alternatives to germane for deposition of Ge-containing films by MOVPE.[10]
Safety edit
Germane is a highly flammable, potentially pyrophoric,[11] and a highly toxic gas. In 1970, the American Conference of Governmental Industrial Hygienists (ACGIH) published the latest changes and set the occupational exposure threshold limit value at 0.2 ppm for an 8-hour time weighted average.[12] The LC50 for rats at 1 hour of exposure is 622 ppm.[13] Inhalation or exposure may result in malaise, headache, dizziness, fainting, dyspnea, nausea, vomiting, kidney injury, and hemolytic effects.[14][15][16]
The US Department of Transportation hazard class is 2.3 Poisonous Gas.[12]
References edit
- ^ a b c d e NIOSH Pocket Guide to Chemical Hazards. "#0300". National Institute for Occupational Safety and Health (NIOSH).
- ^ Yaws, Carl L. (1997), Handbook Of Viscosity: Volume 4: Inorganic Compounds And Elements, Gulf Professional Publishing, ISBN 978-0123958501
- ^ Kunde, V.; Hanel, R.; Maguire, W.; Gautier, D.; Baluteau, J. P.; Marten, A.; Chedin, A.; Husson, N.; Scott, N. (1982). "The tropospheric gas composition of Jupiter's north equatorial belt (NH3, PH3, CH3D, GeH4, H2O) and the Jovian D/H isotopic ratio". Astrophysical Journal. 263: 443–467. Bibcode:1982ApJ...263..443K. doi:10.1086/160516.
- ^ W. L. Jolly "Preparation of the Volatile Hydrides of Groups IVA and VA by Means of Aqueous Hydroborate" Journal of the American Chemical Society 1961, volume 83, pp. 335-7.
- ^ US Patent 4,668,502
- ^ Girolami, G. S.; Rauchfuss, T. B.; Angelici, R. J. (1999). Synthesis and Technique in Inorganic Chemistry. Mill Valley, CA: University Science Books.
- ^ US Patent 7,087,102 (2006)
- ^ a b Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ^ Venkatasubramanian, R.; Pickett, R. T.; Timmons, M. L. (1989). "Epitaxy of germanium using germane in the presence of tetramethylgermanium". Journal of Applied Physics. 66 (11): 5662–5664. Bibcode:1989JAP....66.5662V. doi:10.1063/1.343633.
- ^ Woelk, E.; Shenai-Khatkhate, D. V.; DiCarlo, R. L. Jr.; Amamchyan, A.; Power, M. B.; Lamare, B.; Beaudoin, G.; Sagnes, I. (2006). "Designing Novel Organogermanium MOVPE Precursors for High-purity Germanium Films". Journal of Crystal Growth. 287 (2): 684–687. Bibcode:2006JCrGr.287..684W. doi:10.1016/j.jcrysgro.2005.10.094.
- ^ Brauer, 1963, Vol.1, 715
- ^ a b Praxair MSDS Archived 2012-05-08 at the Wayback Machine accessed Sep. 2011
- ^ NIOSH Germane Registry of Toxic Effects of Chemical Substances (RTECS)accessed Sep. 2011
- ^ Gus'kova, E. I. (1974). "K toksikologii Gidrida Germaniia" [Toxicology of germanium hydride]. Gigiena Truda I Professionalnye Zabolevaniia (in Russian). 18 (2): 56–57. PMID 4839911.
- ^ US EPA Germane
- ^ Paneth, F.; Joachimoglu, G. (1924). "Über die pharmakologischen Eigenschaften des Zinnwasserstoffs und Germaniumwasserstoffs" [About the pharmacological characteristics of tin hydride and germanium hydride]. Berichte der Deutschen Chemischen Gesellschaft (in German). 57 (10): 1925–1930. doi:10.1002/cber.19240571027.
External links edit
- Metaloids (manufacturer) datasheet
- Arkonic Specialty Gases China (manufacturer) datasheet
- Licensintorg Russia (process technology sale) Archived 2013-03-29 at the Wayback Machine
- Honjo Chemical Japan (manufacturer) Archived 2013-01-20 at the Wayback Machine
- Praxair datasheet
- Air liquide gas encyclopedia entry Archived 2013-07-30 at the Wayback Machine
- CDC - NIOSH Pocket Guide to Chemical Hazards
- Voltaix (manufacturer) datasheet Archived 2011-07-17 at the Wayback Machine
- Foshan Huate Gas Co., Ltd. (manufacturer)
- Horst Technologies, Russia (manufacturer)





