
Summary
The Van Leusen reaction is the reaction of a ketone with TosMIC leading to the formation of a nitrile. It was first described in 1977 by Van Leusen and co-workers.[1] When aldehydes are employed, the Van Leusen reaction is particularly useful to form oxazoles and imidazoles.
| Van Leusen reaction | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Named after | Daan Van Leusen Albert M. Van Leusen | ||||||||
| Reaction type | Substitution reaction | ||||||||
| Reaction | |||||||||
| |||||||||
| Identifiers | |||||||||
| Organic Chemistry Portal | van-leusen-reaction | ||||||||
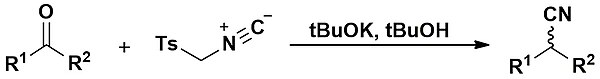
Mechanism edit
The reaction mechanism consists of the initial deprotonation of TosMIC, which is facile thanks to the electron-withdrawing effect of both sulfone and isocyanide groups. Attack onto the carbonyl is followed by 5-endo-dig cyclisation (following Baldwin's rules) into a 5-membered ring.
| Van Leusen oxazole synthesis | |||||||
|---|---|---|---|---|---|---|---|
| Named after | Daan Van Leusen Albert M. Van Leusen | ||||||
| Reaction type | Ring forming reaction | ||||||
| Reaction | |||||||
| |||||||
| Identifiers | |||||||
| Organic Chemistry Portal | van-leusen-oxazole-synthesis | ||||||
If the substrate is an aldehyde, then elimination of the excellent tosyl leaving group can occur readily. Upon quenching, the resulting molecule is an oxazole.
| Van Leusen imidazole synthesis | |
|---|---|
| Named after | Daan Van Leusen Albert M. Van Leusen |
| Reaction type | Ring forming reaction |
| Identifiers | |
| Organic Chemistry Portal | van-leusen-imidazole-synthesis |
If an aldimine is used, formed from the condensation of an aldehyde with an amine, then imidazoles can be generated through the same process.[2]
When ketones are used instead, elimination cannot occur; rather, a tautomerization process gives an intermediate which after a ring opening process and elimination of the tosyl group forms an N-formylated alkeneimine. This is then solvolysed by an acidic alcohol solution to give the nitrile product.
References edit
- ^ Van Leusen, Daan; Oldenziel, Otto; Van Leusen, Albert (1977). "Chemistry of sulfonylmethyl isocyanides. 13. A general one-step synthesis of nitriles from ketones using tosylmethyl isocyanide. Introduction of a one-carbon unit". J. Org. Chem. 42 (19). American Chemical Society: 3114–3118. doi:10.1021/jo00439a002.
- ^ Gracias, Vijaya; Gasiecki, Alan; Djuric, Stevan (2005). "Synthesis of Fused Bicyclic Imidazoles by Sequential Van Leusen/Ring-Closing Metathesis Reactions". Org. Lett. 7 (15). American Chemical Society: 3183–3186. doi:10.1021/ol050852+. PMID 16018616.


